Alzheimer’s is a progressive neurological disorder with classic clinical symptoms such as dementia, cognitive decline, and behavioural changes, particularly in the ageing population. It is characterized pathologically by the accumulation of Amyloid-β (Aβ) plaques and intracellular neurofibrillary tangles (NFT), leading to synaptic dysregulation and neuronal damage. Despite the extensive research, the current treatment primarily focuses on symptom relief, underscoring the need for therapies that can slow down or stop the progression of Alzheimer's disease (AD). This article provides insight into the role of molecular chaperones and their potential as therapeutic targets.

Mechanism of Protein Folding and Aggregation
The precise folding of a protein into a 3D conformation is crucial for its biological function. It is a well-regulated process, but under certain conditions, such as oxidative stress, ageing, genetic mutations, or metabolic dysfunction, these proteins can misfold. The misfolded conformation exposes their hydrophobic residues, which become vulnerable to aggregation.
Tau is a protein that helps microtubules attach to adjacent microtubules. When it undergoes a faulty post-translational modification, such as hyperphosphorylation, the tau protein loses affinity and aggregates into paired helical filaments and neurofibrillary tangles (NFT). Tau pathology disrupts axonal transport, impairs cellular homeostasis, and spreads in a prion-like manner between neurons, contributing to disease progression.
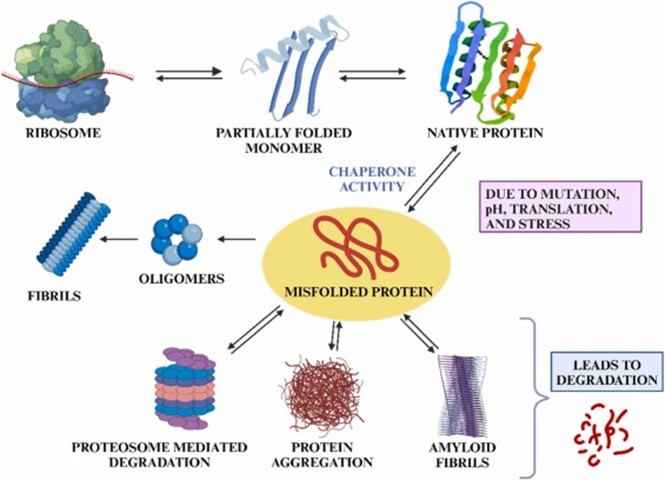
Molecular Chaperons and Protein Quality Control
Molecular chaperons are vital components of the cellular protein quality control system. They assist in protein folding, prevent aggregation, refold misfolded proteins, and target irreversibly damaged proteins for degradation. The majority of proteins belong to the heat-shock protein (Hsp) family, including Hsp70, Hsp90, Hsp60, Hsp40, Hsp100, and small Hsps.
The mode of action of chaperons involves ATP-dependent and ATP-independent mechanisms, in association with co-chaperones, that regulate substrate specificity and folding outcomes. When misfolding occurs, the chaperons direct the proteins to degradation pathways such as the ubiquitin–proteasome system (UPS) or autophagy-lysosome system.
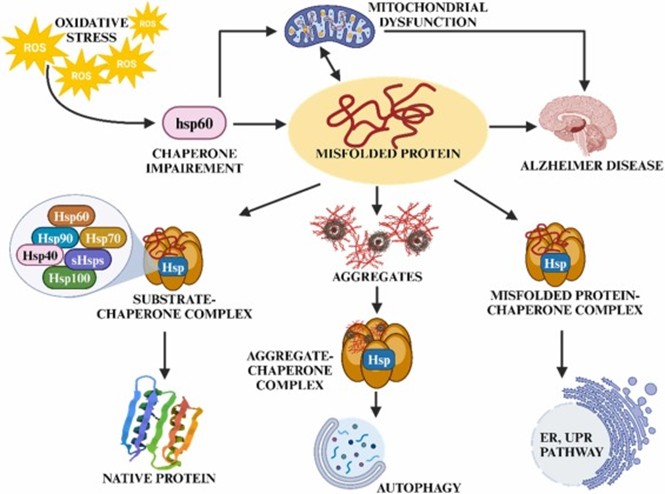
Chaperons Malfunction in Alzheimer’s disease
The dysregulation of Hsp70 and Hsp systems, which are unable to effectively refold or degrade misfolded Aβ or tau proteins, leads to their accumulation. The co-chaperon CHIP, associated with ubiquitination or proteasomal degradation of tau, is adversely affected, which further worsens the tau pathology.
Another co-chaperon, FKBP51, which binds to Hsp 90 and stabilizes the tau conformation. The recent study suggests that elevated FKBP51 levels in AD promote tau hyperphosphorylation and aggregation, making it a potential therapeutic target.
Genetic and Environmental Modulation of Chaperone Function
Alzheimer’s disease (AD) is a multifactorial disorder influenced by genetic and environmental changes, which adversely impact the chaperone system. The study suggests that multiple genes, such as APP, PSEN1, PSEN2, and APOE ε4, are responsible for early- and late-onset AD, respectively. Genome sequencing also identified additional genes involved in amyloid processing, trafficking, and proteostasis, related to the chaperone-dependent pathway.
Environmental factors such as air pollution, pesticides, nanoparticles, and heavy metals create oxidative stress, which further overwhelms the chaperone mechanism, leading to an increased risk of AD.
Chaperone-Targeted Therapeutic Strategies
Molecular chaperones play a central role in proteostasis and represent potential therapeutic targets in AD. Several approaches are under investigation-
(i) Small-molecule chaperone modulators
The chaperon molecule, Hsp 70, is identified to play a crucial role in promoting tau degradation, reducing Aβ accumulation, which further facilitates proteasomal clearance. For geranylgeranylacetone, celastrol, YM-01, and MKT-077. Additionally, selective inhibition of Hsp90 by geladanamycin shows promising results in preclinical trials.
(ii) Co-Chaperons mediated pathways
FKBP51 is a co-chaperone of Hsp90 that supports the folding and function of associated proteins such as transcription factors and kinases. Wang et al. identified potent inhibitors of the FKBP51–Hsp90 interaction using structure-based virtual screening. Additionally, the small molecule LA1011 disrupts this interaction, thereby providing neuroprotection.
(iii) Gene Therapy
Gene therapy approaches based on targeted overexpression of protective chaperones in the brain support the clearance of misfolded proteins. The BRICHOS domain, a specialized anti-amyloid chaperone, has demonstrated potent inhibition of Aβ42 oligomer formation and neurotoxicity in vitro and in vivo. Viral delivery of BRICHOS or CHIP has shown beneficial effects in animal models, improving protein homeostasis and cognitive outcomes.
Challenges and Future Perspective
Despite recent advances and encouraging preclinical results, translating chaperone-based therapies into clinical success remains challenging. AD’s complexity, heterogeneity, and limitations of current disease models hinder effective translation. Moreover, chaperones regulate numerous cellular pathways, raising concerns about off-target effects and safety.
Advances in high-throughput screening, omics technologies, and improved disease models are accelerating the identification of selective chaperone modulators. Comprehensive chaperone-targeted therapies, combined with other approaches such as immunotherapy or metabolic interventions, may yield more effective treatments.



Discussion