Introduction
Atherosclerotic cardiovascular diseases (ASCVDs) remain the leading cause of death worldwide, contributing to approximately 17.9 million to 19.8 million deaths worldwide annually (Tran et al., 2026). Within the United States alone, as many as 30% of all deaths annually are attributed to ASCVDs. As the name suggests, the major underlying cause of ASCVDs is the progression of atherosclerosis, a chronic disease characterized by the hardening of the arterial walls of major blood vessels due to the accumulation of low-density lipoproteins (LDLs) within them.
Cholesterol Carriers Form Fatty Streaks that Lead to Atherosclerosis
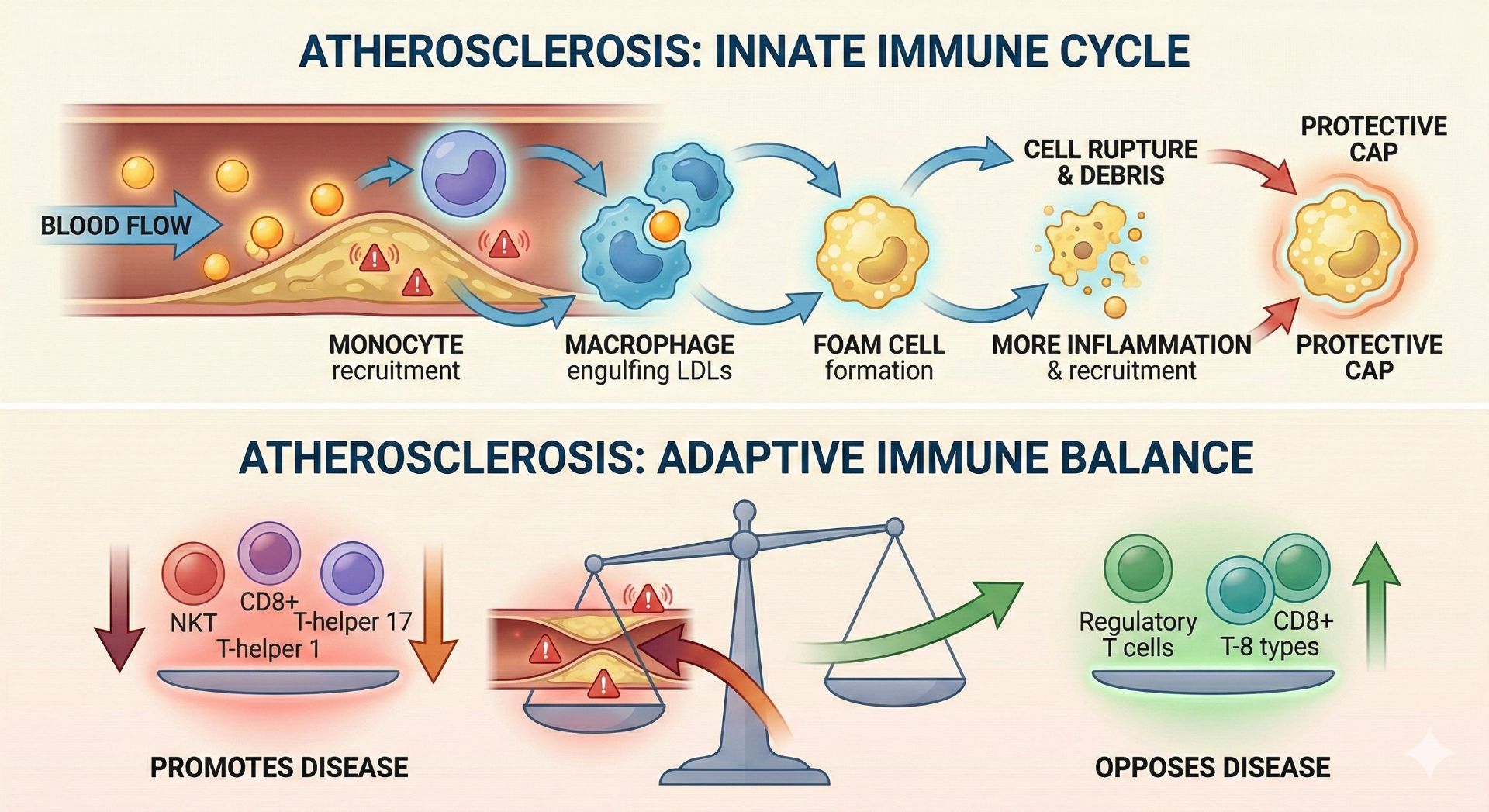
LDLs are the major carrier proteins in the body responsible for transporting cholesterol through the blood to cells within the body that need it. The LDLs make their way inside the blood vessel walls when the innermost layer of cells, the endothelia, are subjected to continuous shear stresses and toxins, resulting in chronic injury. The damage sustained by the endothelia ultimately allows the LDLs to penetrate through it, where they will begin to accumulate. Once inside the arterial walls, the LDLs will undergo various types of modifications, including oxidation, which will convert them into modified LDLs (mLDLs) and trigger an immune response by the endothelial cells, which will release chemokines to attract monocytes, dendritic cells, and neutrophils to the site of injury (He et al., 2024).
Innate Cells are First Responders to Atherosclerotic Plaques
The neutrophils will typically reach the damaged tissue first, where they will secrete reactive oxygen species (ROS) and proteases to aggravate damage to the endothelia and make it easier for other immune cells to pass through the arterial walls. They also work alongside dendritic cells, macrophages, and neutrophils as antigen-presenting cells (APCs), which are important in stimulating cells and initiating the adaptive immune response. Most importantly, they stimulate macrophages, which increases their mLDL uptake, making them more prone to becoming foam cells.
Besides neutrophils, monocytes will enter the arterial wall and become resident tissue macrophages. Macrophages are a type of phagocytic cell that play a major role in the clearance of pathogens and debris from tissue. These newly formed macrophages will then recognize the mLDLs as debris that needs to be cleared from the arterial wall and will begin to engulf the mLDLs through a process known as endocytosis. Over an extended period of time, the macrophages will eventually consume too many mLDLs, which causes them to become foam cells. At this point, they become unstable and capable of rupturing (Gui et al., 2022). To prevent widespread damage caused by the potential rupturing of the foam cells, a protective fibrous cap will begin to form around them, and additional immune cells from the blood will be recruited.
This results in the release of partially digested mLDL, cholesterol, and macrophage cellular debris, which in turn results in a more severe immune response.
Diseased Macrophages Activate the Adaptive Immune System
As the fibrous cap continues to form around the vascular smooth muscle cells (VSMCs) and saturated macrophages, inflammation signaling begins through the release of proinflammatory cytokines such as interferon gamma (IFN-γ), tumor necrosis factor alpha (TNF-α), and interleukin-1beta (IL-1β). These inflammatory molecules trigger the release of matrix metalloproteinases (MMPs) from VSMCs within the arterial walls, making it easier for other immune cells from the blood to enter (Liu et al., 2024). As a result, although monocytes and macrophages play a major role in the early stages of atherosclerosis, in the later stages, the disease becomes more immunologically complex.
More innate cells are continuously recruited to the injured tissue, where many of them will serve as APCs, which are necessary to activate adaptive immune cells, primarily consisting of B lymphocytes and T lymphocytes. Some resident B cells, which are known as adventitial B cells, will work alongside the other APCs to stimulate non-resident B cells and T cells. Activated B cells will either promote disease progression by becoming B2 cells or will oppose it by becoming B1 cells.
Several types of T cells are also involved in later stages of atherosclerosis, including natural killer T (NKT) cells, CD8+ T cells, and CD4+ T cells, which include regulatory T cells and all different types of helper T cells. Some activated T cells will contribute to atherosclerotic disease, such as some NKT cells, some CD8+ T cells, T-helper 1 cells, some T-helper 2 cells, and some T-helper 17 cells. On the other hand, some T cells will protect against atherosclerotic progression, including other NKT cells, other CD8+ T cells, regulatory T cells, other T-helper 2 cells, and other T-helper 17 cells.
What Can You Do?
The key takeaway here is that although the immune system plays an instrumental role in the development and progression of ASVCDs, it does not act alone. While atherosclerosis is a natural part of the aging process for most people, its severity can be mitigated by a variety of external factors, including a healthy diet and exercise, both of which substantially decrease the number of LDLs that circulate through the bloodstream and limit the exposure of endothelial walls to chronic injury. If you have a history of CVDs in your family, work with a doctor, dietitian, or nutritionist to come up with a heart-healthy plan that works best for you. Not everyone’s bodies are the same.
Conclusion
The immune system is heavily involved in the progression of atherosclerosis. Its role in the disease is complex, with several immune cells contributing to disease progression, and other immune cells working against disease progression. The major driving force behind the formation of atherosclerotic plaque formation is chronic injury of the inner cell wall of the major blood vessels (the endothelia), prompting cholesterol-carrying LDL proteins to accumulate inside the endothelial walls, where they undergo chemical modifications that make them immunoreactive. This results in the recruitment of both innate and adaptive immune cells, foam cell formation, inflammation, and arterial wall hardening (due to the eventual calcification of the rupturing plaques), narrowing,g and instability. At this point, the disease can become life-threatening and result in various forms of ASVCDs, which may include coronary heart disease (CHD), cerebrovascular disease, peripheral artery disease, and aortic atherosclerotic disease.




Discussion