Introduction
Vancomycin is an essential glycopeptide antibiotic used to treat life-threatening infections caused by Gram-positive bacteria, particularly Methicillin-Resistant Staphylococcus aureus (MRSA). It functions by inhibiting bacterial cell-wall synthesis and is still the most potent drug to be used when others fail (Patel, Preuss, & Bernice, 2023). Similar to maintaining the right temperature in an incubator, dosing vancomycin needs to be done very carefully. The desired effect won't take place if the temperature is too low, and the environment will become harmful if the temperature is too high. In the same way, not enough vancomycin exposure lets the infection persist, but too much increases the chance of kidney toxicity.
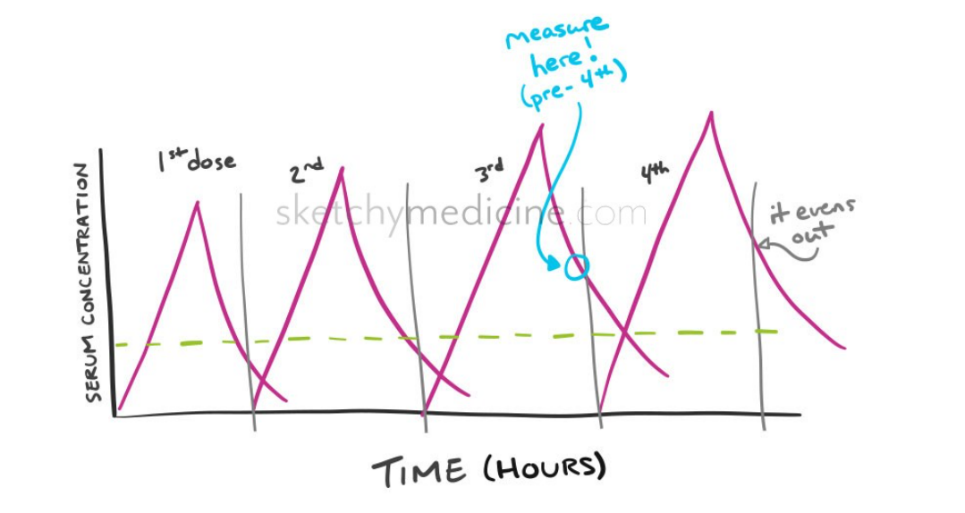
For a long time, vancomycin dosing was mainly based on tracking trough levels, i.e., the lowest concentration of the drug before the next dose. The assumption was simple: if the trough stayed within a certain range, the patient was safe and the treatment effective. However, now there is evidence that reveals that this method is not perfect at all.
Currently, a more accurate and scientific method is taking the lead: AUC-guided monitoring. It measures the total exposure of the drug in the bloodstream over time and thus gives a more comprehensive insight into the dosing decisions (Ashley Shiyuan Lim et al., 2023).
This article discusses the meaning of AUC-guided monitoring, its superiority over traditional trough measurements, and the impact of AUC monitoring on antibiotic therapy delivery by healthcare professionals.
1. Why Traditional Trough Monitoring Falls Short
Trough monitoring was once the gold standard. Doctors targeted a trough level between 15 and 20 mg/L, hypothesizing that this interval reflected the total drug exposure necessary to combat infection (Am J Health-Syst Pharm, 2009)
However, multiple studies began to question this viewpoint. Research showed that many patients with “therapeutic” trough levels still developed kidney toxicity, while others with lower troughs successfully cleared their infections (Neely et al., 2018; Finch et al., 2017). The problem is that a single trough value only refers to one point in time. It is unable to represent how much drug a patient has been given during the day.
According to the 2020 American Society of Health-System Pharmacists (ASHP) and Infectious Diseases Society of America (IDSA) guidelines, trough-based dosing is not sufficient. They advocated for the use of Area Under the Curve (AUC) monitoring that is more closely linked to both effectiveness and safety (Rybak et al., 2020).
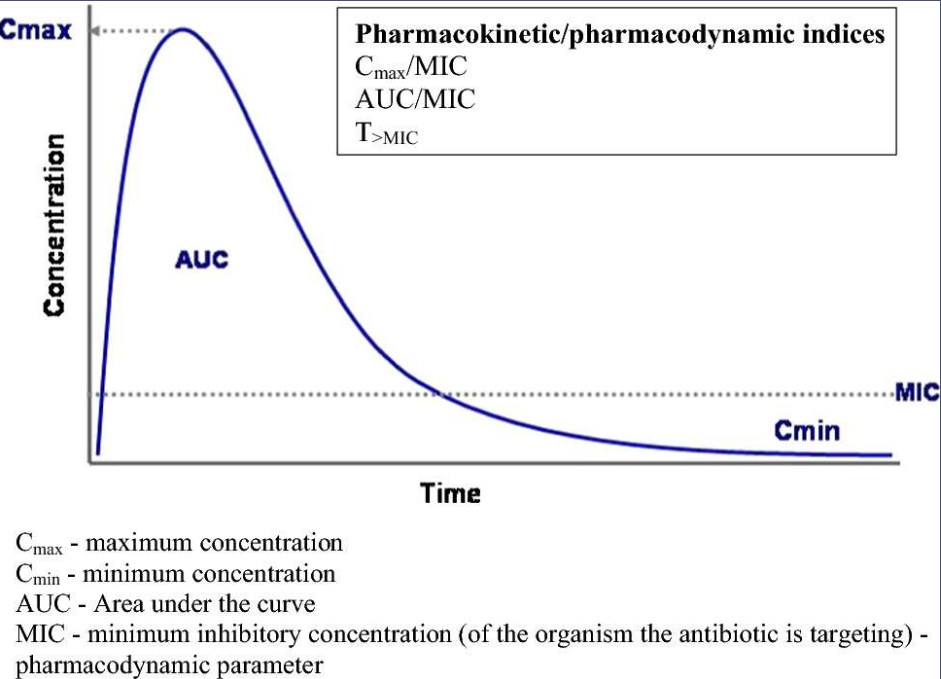
2. Understanding the AUC Concept
AUC, or Area Under the Concentration–Time Curve, is a measure of the total drug exposure in a patient’s blood over 24 hours. It gives physicians information about how much drug the body has handled instead of how much drug is left just before the next dose.
The target AUC/MIC ratio for vancomycin (where MIC stands for Minimum Inhibitory Concentration) is 400 to 600 mg·h/L (Du Sault et al., 2022).
Below 400, the infection may not respond to treatment.
Above 600, the patient faces a higher risk of kidney damage.
You can think of this like sunlight shining on solar panels. It is not about the brightest moment of the day or the dimmest one, but the total amount of sunlight that hits the panels over time. In the same way, AUC focuses on overall exposure instead of isolated snapshots, which offers a more accurate picture of the drug’s effect inside the body.
The table below summarises the difference between traditional trough monitoring and AUC-guided dosing:
Feature | Trough-Based Monitoring | AUC-Based Monitoring |
|---|---|---|
Monitored Value | The minimum drug concentration (Cmin) in the blood, just before the next dose is administered. | The total drug exposure over a dosing interval, calculated as the Area Under the Concentration-Time Curve (AUC). |
Sampling | Requires a single blood sample drawn at a specific time point (just before the next dose). | Typically requires multiple blood samples (e.g., peak and trough) or advanced Bayesian software modelling with fewer samples to estimate the entire curve. |
Visual on a Graph | A single point at the lowest part of the curve. | The entire area beneath the concentration-time curve for one dosing period. |
Goal | Ensure the minimum concentration is above a certain threshold for efficacy and below a toxicity limit (though this method has proven less reliable for toxicity). | Ensure the total drug exposure (AUC) falls within a specific therapeutic range for optimal efficacy and safety, often associated with a lower risk of kidney injury (nephrotoxicity). |
Complexity | Simpler to implement in clinical practice. | More complex, often requiring specialised software or calculations. |
These differences highlight why AUC-guided monitoring provides a more individualized understanding of vancomycin exposure which makes it the preferred method in modern practice.
3. How Clinicians Measure AUC
There are two main approaches used in hospitals today.
A. The Two-Point Pharmacokinetic Method
This method involves taking two blood samples: one shortly after infusion (the peak) and the other just before the next dose (the trough). The values are computed into pharmacokinetic formulas to determine total exposure in 24 hours. It is effective but labour-intensive and not suitable for a large number of patients (Liu et al., 2022).
B. Bayesian Modelling
This state-of-the-art technique requires software that integrates a patient’s personal information, e.g. age, weight, kidney function, and infection, with a large pharmacokinetic library to provide a predicted AUC. Examples of such tools include InsightRX, PrecisePK, and DoseMeRx, which provide dosing suggestions along with AUC calculations, requiring fewer blood draws (Brocks and Hamdy, 2020). For example, if a young trauma patient clears drugs too quickly due to increased kidney function, the model recommends a higher or more frequent dose. On the other hand, an elderly patient with impaired kidney function will be given a lower dose. Such flexibility makes AUC-guided therapy very efficient and personalised.
4. Advantages and Outcomes of AUC-based Monitoring
A. AUC-guided monitoring is associated with significant reductions in vancomycin toxicity and unnecessary dose escalation. This is achieved even without an increased risk of acute kidney injury (Robinson et al., 2023).
B. Implementation is more practical with Bayesian approaches. This method requires fewer samples and allows flexible timing. This makes AUC-guided dosing feasible in resource-limited hospitals and even in outpatient settings where blood collection may be less frequent (Gillett et al., 2024).
C. Successful adoption depends on interdisciplinary teamwork, ongoing staff education, and strong informatics support to ensure accurate and consistent application of AUC-based protocols (Finch et al., 2017).
Overall, integrating AUC-guided vancomycin monitoring has led to safer drug use and more effective therapy across clinical settings. The growing role of digital health tools, coupled with pharmacist leadership, continues to drive progress toward standardised and data-driven antimicrobial dosing.
5. Bringing AUC Monitoring into Practice
Implementing AUC-guided monitoring requires collaboration between pharmacists, physicians, and laboratory teams. The typical process revolves around obtaining one or two timed vancomycin levels, inputting them into validated software, and then following the dose adjustment recommendations.
While the procedure may be complicated in the beginning, several hospitals have reported better patient outcomes and lower adverse events after the implementation of AUC-based protocols. Further, improvements in the digital medical record system and AI-assisted pharmacokinetic tools are facilitating bedside AUC calculation even more.
Conclusion
Changing from trough-based to AUC-guided vancomycin monitoring is a big leap in precision medicine. A single test is no longer relied on by clinicians, and now they can make dosing decisions based on a complete understanding of drug behavior in the body. This method improves safety, boosts treatment success, and reflects a broader transformation in healthcare.
For students and early-career scientists, comprehending the functioning of AUC-guided monitoring is far beyond just understanding pharmacokinetics. It is to recognize how the interconnection of science, technology, and patient care results in safer and smarter medicine.



Discussion