Introduction
Biologics have transformed the management of chronic and life-threatening diseases such as cancer, autoimmune disorders, and metabolic conditions. However, their complexity, high production costs, and limited affordability continue to challenge healthcare systems worldwide. (Vishwakarma, Pushkar, et al.) As patents for many blockbuster biologics expire, biosimilars have emerged as reliable and cost-effective alternatives that maintain the same therapeutic value. Developed through rigorous analytical and clinical comparisons, biosimilars offer a path toward wider treatment access without compromising quality. (Ho Cynthia M) This article discusses the growing relevance of biosimilars, the factors driving their expansion, and the evolving landscape of modern biologic therapy.
Biosimilars: Need, Concept, and Growth Drivers
The need for biosimilars stems from the increasing dependence on biologic therapies and the financial burden they impose. Many pioneering biologics such as adalimumab, trastuzumab, and bevacizumab have reached the end of their exclusivity period, creating opportunities for equally effective but more affordable alternatives. A biosimilar is not an identical copy but a highly similar version of a reference biologic, requiring extensive characterization, pharmacokinetic comparisons, and clinical evaluations to confirm similarity in safety, purity, and potency. (Rompas, Sotiris, et al. ) Their accelerated global adoption is driven by several key factors: the rising healthcare costs associated with biologics, strong regulatory frameworks established by agencies like the FDA and EMA, and increasing real-world evidence validating biosimilar safety and effectiveness. Together, these elements have positioned biosimilars as essential contributors to more sustainable healthcare systems. (Mascarenhas-Melo, Filipa, et al.)
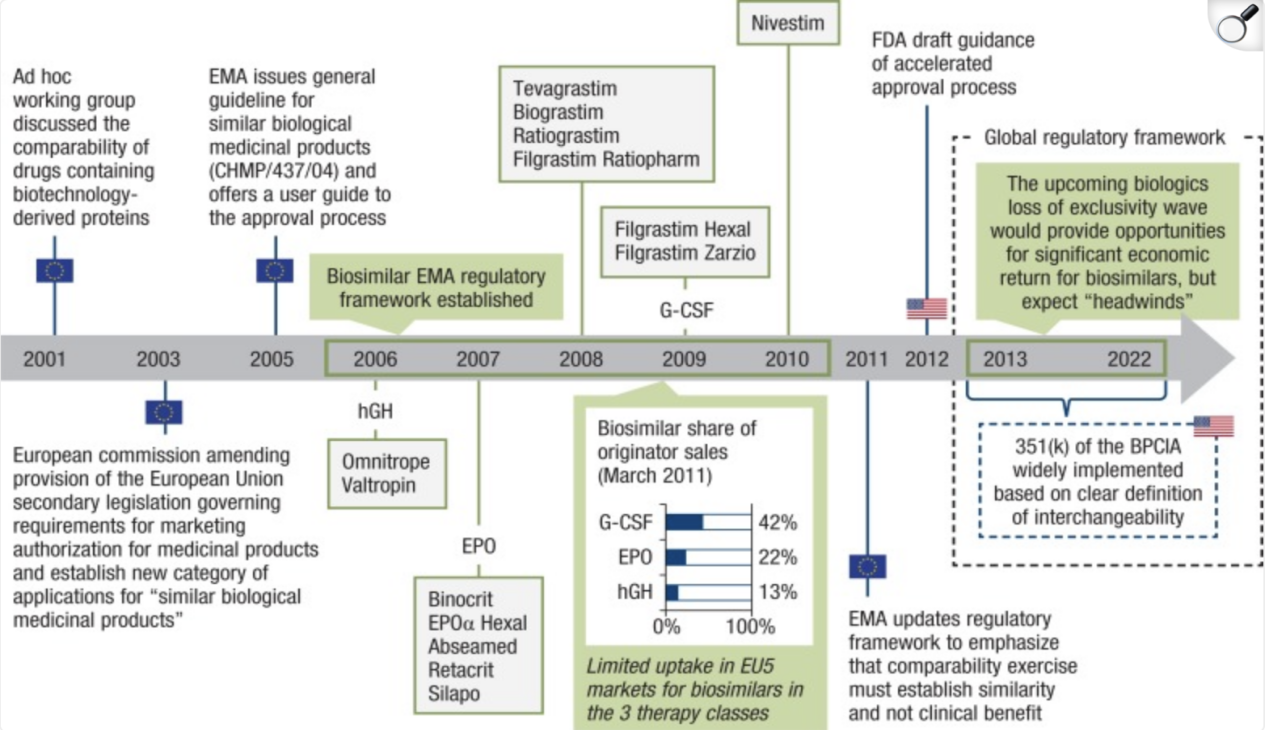
Impact on Healthcare and Market
Biosimilars have significantly influenced both clinical practice and global pharmaceutical markets. In many therapeutic areas including oncology, rheumatology, endocrinology, and ophthalmology biosimilars provide treatment outcomes comparable to originator biologics, offering physicians confidence in switching or initiating therapy. Their introduction has widened patient access, particularly in regions where high biologic prices previously restricted availability. (Tsiftsoglou et al.)
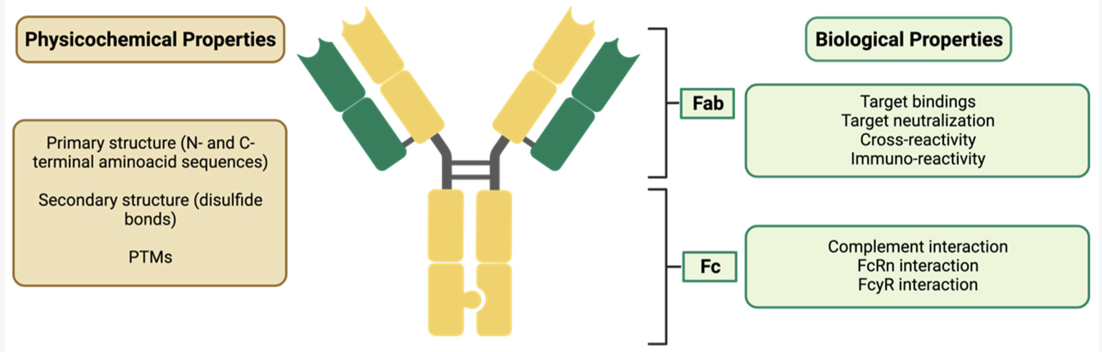
Economically, biosimilars help reduce overall healthcare expenditure. Countries with early biosimilar adoption, especially in Europe, have reported substantial savings that can be reinvested in advanced therapies, diagnostics, and patient services. Market forecasts indicate consistent growth, with the global biosimilar sector expected to expand rapidly as more biologics lose patent protection and prescriber acceptance continues to rise. (Troein, P., et al.)
Challenges and Future Outlook
Despite their advantages, biosimilars face several challenges. Manufacturing remains highly complex, requiring sophisticated bioprocessing equipment, stringent quality control, and skilled scientific expertise. Patent litigation and regulatory delays can slow market entry, while misconceptions among patients or providers may affect acceptance. Ensuring robust pharmacovigilance and post-marketing surveillance is also essential to maintain trust.
Looking ahead, the future of biosimilars is promising. Continued development of next-generation biosimilars, integration of AI-based bioprocess optimization, and increased harmonization of global regulatory standards are expected to streamline development and improve accessibility. As technological capabilities grow, biosimilars will likely expand into more complex biologic categories, further strengthening their role in healthcare. (Kaida-Yip, Flyn, et al.)
Conclusion
The expanding presence of biosimilars reflects a pivotal shift toward more accessible, affordable, and sustainable healthcare. With strong scientific validation, favourable regulatory support, and growing clinician confidence, biosimilars are positioned to play a critical role in meeting global therapeutic needs. Their continued growth will not only enhance treatment accessibility but also reinforce the balance between innovation and affordability—shaping a more equitable future for biologic medicine.



Discussion