Why oxygen reactions matter
Selective oxidation reactions are central to modern biochemistry. They enable the controlled introduction of oxygen into organic molecules, a key step in the synthesis of pharmaceuticals and fine chemicals. However, many of the enzymatic systems traditionally used for these transformations are complex and difficult to implement in practice. Peroxygenases have recently emerged as a simpler alternative, as they use hydrogen peroxide directly as the oxygen source and do not require auxiliary proteins or costly cofactors. In this article, we describe what peroxygenases are, how they catalyze oxidation reactions, and why they represent an increasingly relevant class of enzymes for modern and more sustainable biochemistry.
What are peroxygenases, and how do they work?
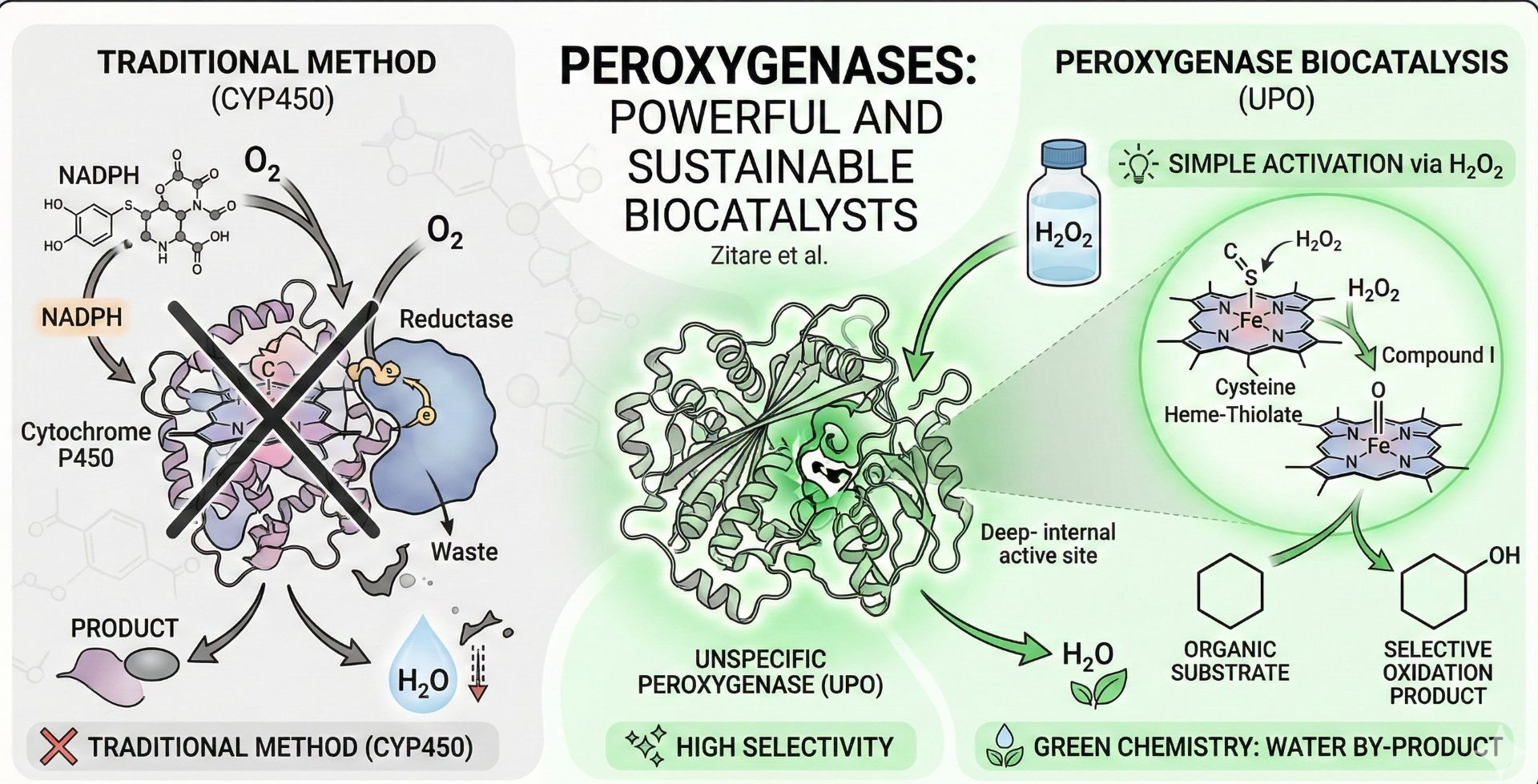
Peroxygenases are enzymes that use hydrogen peroxide as a direct source of oxygen to carry out oxidation reactions. In simple terms, they add an oxygen atom to other molecules in a precise and controlled way. They belong to a group called heme enzymes, which means they contain a small iron-based group (heme) that helps activate oxygen inside the protein.
What makes peroxygenases especially interesting is that they can perform reactions similar to those of more complex oxidation enzymes, but in a much simpler way. They use hydrogen peroxide directly and do not need extra helper proteins or complicated reaction systems. This makes their reactions easier to control and easier to apply in real laboratory and industrial settings.
Why are they important in modern biochemistry?
Peroxygenases are important because they offer a simpler and more sustainable way to carry out selective oxidation reactions. They reduce the need for complex enzyme systems, extra proteins, and expensive cofactors, making biocatalytic processes easier to design and operate.
In industrial and pharmaceutical chemistry, their ability to selectively modify complex molecules makes them useful for producing valuable intermediates for drugs and fine chemicals. In addition, because peroxygenases use hydrogen peroxide and mainly generate water as a by-product, they are increasingly seen as practical and environmentally friendly alternatives for scalable and sustainable oxidation processes.
Data and evidence from recent studies
Experiments show that enzymes related to peroxygenases can produce reactive forms of oxygen in a simple and efficient way. For example, a recent study by Zitare and co-workers showed that dye-decolorizing peroxidases (DyPs) can be activated by reactive oxygen molecules to start their catalytic activity, without needing complex supporting systems. When the researchers supplied small and controlled amounts of superoxide or hydrogen peroxide, the enzymes worked as well as, or even better than, in standard reactions.
An important and easy to understand message from this work is that not all enzymes behave in the same way. Some respond better to superoxide, while others work best with hydrogen peroxide. This means that each enzyme is tuned to its own structure. The study also showed that a single amino acid located near the heme group plays a key role in guiding reactive oxygen to the iron center and in keeping the enzyme active during the reaction (Zitare et al., 2024).
These results show in a direct way that small changes around the heme group can strongly influence how hydrogen peroxide is activated and how oxygen is transferred during a reaction. This idea is also central to understanding how fungal peroxygenases work.
Further evidence comes from mutation and spectroscopic studies on an ancestral fungal DyP enzyme. Zitare and co-workers showed that two amino acids located close to the heme group, an aspartate and an arginine, are essential for the efficient use of hydrogen peroxide and for the internal movement of electrons within the protein. When these amino acids were changed, the enzyme lost most of its activity. This shows that the exact shape of the active site is crucial for how well the enzyme uses hydrogen peroxide and transfers oxygen to other molecules (Zitare et al., 2020).
An important recent study by Zitare and co-workers demonstrates in a clear way that the local environment around a protein can strongly affect how it binds oxygen. Using a sensitive spectroscopic method, the authors studied myoglobin placed on surfaces that mimic biological membranes. They found that even brief and weak contacts with these surfaces slightly change the shape of the region where oxygen binds. As a result, the bond between iron and oxygen becomes weaker, and the protein holds oxygen less tightly (Zitare et al., 2026).
Although this study focuses on myoglobin and not directly on peroxygenases, it clearly shows that small changes around the heme group can alter how a protein binds to other molecules and how it carries out chemical reactions. This is especially relevant for peroxygenases, as their activity depends on precise control of hydrogen peroxide activation and the shape of the active site. Together with previous studies on DyP enzymes, shows that the local environment of a heme-containing enzyme decisively influences the chemistry of oxygen and peroxide, a key aspect for improving and designing new peroxygenases for biocatalytic applications.
Practical implications
Peroxygenases make selective oxidation reactions easier to perform and easier to apply in real-world settings. Because they can insert oxygen into molecules with high precision, they can be used to prepare useful compounds such as drug intermediates, fine chemicals, and modified natural products used in fragrances and pharmaceuticals.
Compared with classical cytochrome P450 systems, peroxygenases are much simpler to use. They work without extra partner proteins or expensive cofactors, which means simpler reaction setups and easier scale-up.
Peroxygenases are also attractive from an environmental point of view. They use hydrogen peroxide and mainly produce water as a by-product, helping to reduce waste and the use of hazardous chemicals. For this reason, they represent a promising and inspiring step toward cleaner and more sustainable biocatalysis. With further improvements in stability and performance, peroxygenases may soon become key tools for future green oxidation chemistry, bringing high-precision enzyme catalysis closer to everyday industrial reality.


Discussion