For the last century, medicine relied on "small molecules"—static chemicals like aspirin or penicillin that march into the body to block a receptor or kill a bacterium. But we have entered a new era. We are no longer just treating symptoms; we are rewriting the biological code that causes them.
Cell and gene therapies represent a seismic shift in healthcare. They promise to cure the incurable, restoring sight to the blind and erasing cancer from the blood. But this frontier is wild. It comes with biological price tags, potential genetic misfires, and a regulatory landscape that is still being mapped.
Are we looking at the holy grail of medicine, or are we rolling the dice with human biology?
Living Drugs: Not Your Grandmother’s Aspirin
To understand the stakes, we must understand the tool. Traditional drugs are chemically defined and constant. Cell therapies, however, are "living drugs." As the Harvard Stem Cell Institute explains, these treatments involve transferring intact, live cells into a patient to help lessen or cure a disease.¹
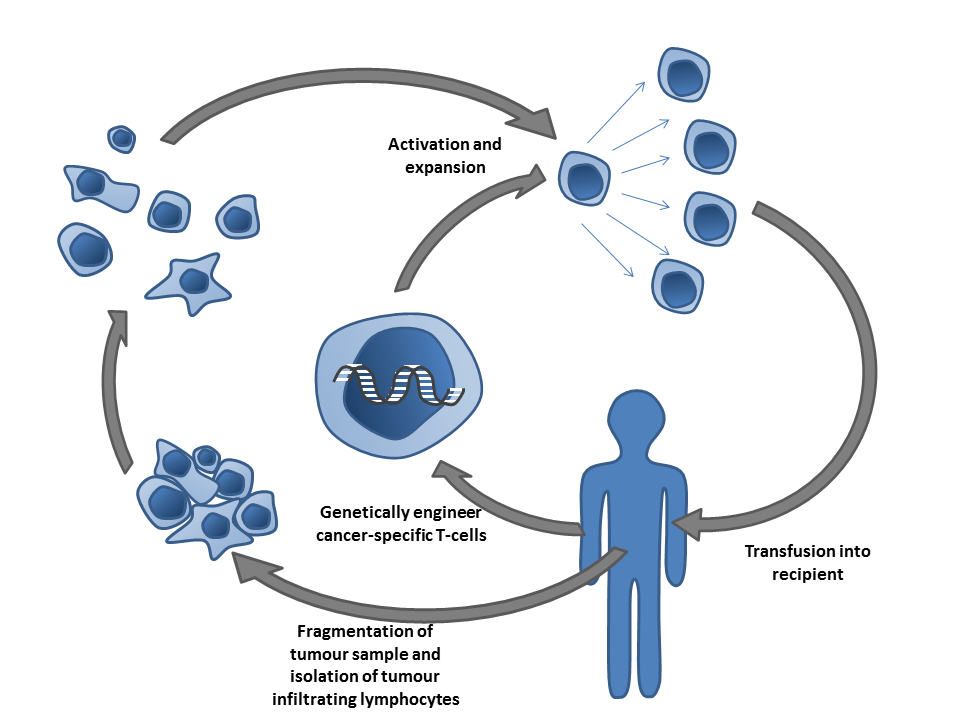
There are two primary ways this biological swap happens:
Autologous therapy: Doctors harvest your own cells, engineer them in a lab to fight disease, and infuse them back into your body. This is the ultimate personalized medicine—a custom suit tailored to your genetic makeup to minimize rejection.
Allogeneic therapy: Cells are taken from a healthy donor and used to treat other people. Think of this as "off-the-shelf" cell therapy, which is scalable but carries higher risks of immune rejection.¹
Often, this involves gene therapy: modifying the DNA within those cells. We might replace a missing gene, like a mechanic replacing a spark plug, or inactivate a mutated gene that’s causing trouble.²
The "Cure Genes": When Science Fiction Becomes Fact
The enthusiasm surrounding these therapies isn't just hype; it’s backed by results that border on miraculous.
The Cancer Hunter
CAR T-Cell Therapy Perhaps the biggest success story lies in the treatment of blood cancers. CAR T-cell therapy involves extracting T-cells (the immune system’s soldiers) and genetically engineering them to produce a specific surface structure called a Chimeric Antigen Receptor (CAR).³
This receptor acts like a GPS tracker. When these engineered cells are returned to the patient, the CAR allows them to hunt down and bind to specific proteins (antigens) on the surface of cancer cells, destroying them. The FDA has approved multiple products, such as Kymriah (tisagenlecleucel) and Yescarta (axicabtagene ciloleucel), for specific lymphomas and leukemias that previously had no hope.⁴
Fixing the Broken Blueprint
Beyond cancer, these therapies are tackling genetic disorders. The University of Utah highlights the success in treating Severe Combined Immune Deficiency (SCID)—often called "Bubble Boy Disease." By introducing a functional gene into the stem cells of these children, their immune systems can rebuild themselves, allowing them to leave isolation and live normal lives.⁹
We are also seeing victories in vision. The FDA-approved therapy Luxturna treats patients with a specific form of inherited retinal dystrophy caused by mutations in the RPE65 gene. It delivers a normal copy of the gene directly into retinal cells, restoring vision in patients who were going blind.⁵ ⁶
The Risky Gamble: When Biology Fights Back
If the rewards are high, the risks are equally steep. Messing with the building blocks of life is not without consequence.
The Cytokine Storm
The most immediate danger in therapies like CAR T-cell is a phenomenon called Cytokine Release Syndrome (CRS). When these super-charged T-cells attack cancer, they release massive amounts of inflammatory chemicals called cytokines into the blood. It’s an immune system overdrive. Patients can suffer high fevers and dangerous drops in blood pressure. In severe cases, this "storm" can be fatal.³
The Genetic Wild West: Insertional Mutagenesis
There is also the risk of "off-target effects." When we use viral vectors (modified viruses) to deliver genetic material, we can’t always control exactly where that gene lands in the DNA.
According to a review in Genes & Diseases, there is a theoretical risk of tumorigenicity (causing tumors). If the new gene inserts itself next to an oncogene (a gene that can cause cancer), it could accidentally switch it on. This is known as insertional mutagenesis. While newer technologies are increasing precision, the fear of inadvertently causing a secondary cancer while trying to cure the primary one remains a significant hurdle.⁸
The Solid Tumor Wall
While cell therapies have decimated liquid tumors (blood cancers), they struggle against solid tumors (like breast or lung cancer). A review in Signal Transduction and Targeted Therapy notes that solid tumors create a hostile microenvironment that suppresses the immune system and prevents T-cells from penetrating the mass. Cracking this code is the next great challenge for researchers.⁷
Navigating the Unknown: Regulation and Cost
Science is moving faster than the rulebook. Because these products are "medical products of human origin," they don't fit the standard manufacturing mold of a pill factory.
The Supply Chain Tightrope
The NHS Blood and Transplant service notes that the logistics are incredibly complex. We are dealing with living material that must be collected, processed, and transported at specific temperatures without contamination. This isn't just shipping boxes; it's a "vein-to-vein" supply chain that requires rigorous traceability standards.¹⁰
The Price of Life
Furthermore, the economic barriers are staggering. These treatments are astronomically expensive, often costing hundreds of thousands of dollars per infusion. As noted in recent academic reviews, the high cost of manufacturing, combined with the personalized nature of autologous therapies—creates a massive accessibility gap. If a cure exists but is only available to the ultra-wealthy or those with specific insurance, have we truly succeeded?⁷
So What? The Future of Your Health
What does this mean for you?
1. The end of "one size fits all": Medicine is becoming hyper-personalized. We are moving toward a future where treatments are grown from your own biology, not picked from a generic shelf.
2. A shift from treatment to cure: We are moving away from managing chronic diseases (taking a pill every day forever) to potential "one-and-done" interventions that fix the root cause.
3. Caution is key: If you or a loved one is considering a clinical trial for cell therapy, understand that "approved" does not mean "risk-free." Ask specifically about CRS and long-term monitoring for secondary malignancies.
Conclusion
Cell and gene therapies are neither pure magic nor reckless gambling; they are the calculated, high-stakes evolution of medicine. We have successfully transitioned from the tragedies of early trials to FDA-approved cures for blindness and leukemia.
The risks, from immune storms to genetic misfires, are real, but they are becoming more manageable as our technology refines. We stand on the precipice of a world where "incurable" is just a temporary label. The gamble is high, but for a child regaining their sight or a cancer patient reclaiming their life, it is a bet worth making.




Discussion