Introduction
Animal models have traditionally served as the cornerstone of drug safety evaluation; however, major translational challenges persist due to interspecies differences in physiology, metabolism, and genetic regulation. Many compounds demonstrating favorable toxicity profiles in animals later fail during human trials or are withdrawn post-marketing due to unforeseen adverse effects, especially hepatotoxicity and cardiotoxicity (Lee et al., 2025). Coupled with ethical concerns and regulatory adoption of the 3Rs principle (Replacement, Reduction, Refinement), these limitations have accelerated the shift toward human-relevant in-vitro platforms. Organoids, self-organizing, three-dimensional cellular structures derived from pluripotent stem cells or adult tissue stem cells offer a physiologically relevant alternative by recapitulating tissue-specific architecture, cell heterogeneity, and functional phenotypes more accurately than conventional 2D cell lines (Matsui & Shinozawa, 2021).
Organoid Technology and Its Role in Toxicology
Organoids mimic in-vivo microenvironmental cues, including extracellular matrix signaling, cell-cell interactions, and biochemical gradients. As a result, they reproduce organ-specific metabolism, transport mechanisms, and stress responses critical for toxicity assessment. Compared to animal models, organoids capture human-specific drug responses, enabling more accurate prediction of therapeutic windows and adverse events (Mukhopadhyay & Paul, 2023). Recent engineering advances—such as CRISPR-based gene modification, defined hydrogel matrices, microfluidic perfusion platforms, and 3D-bioprinting—have enhanced organoid maturation, vascular-like perfusion, and functional complexity (Yao et al., 2024). These innovations enable long-term, dynamic drug exposure studies and real-time monitoring of metabolic responses, significantly improving toxicological relevance.
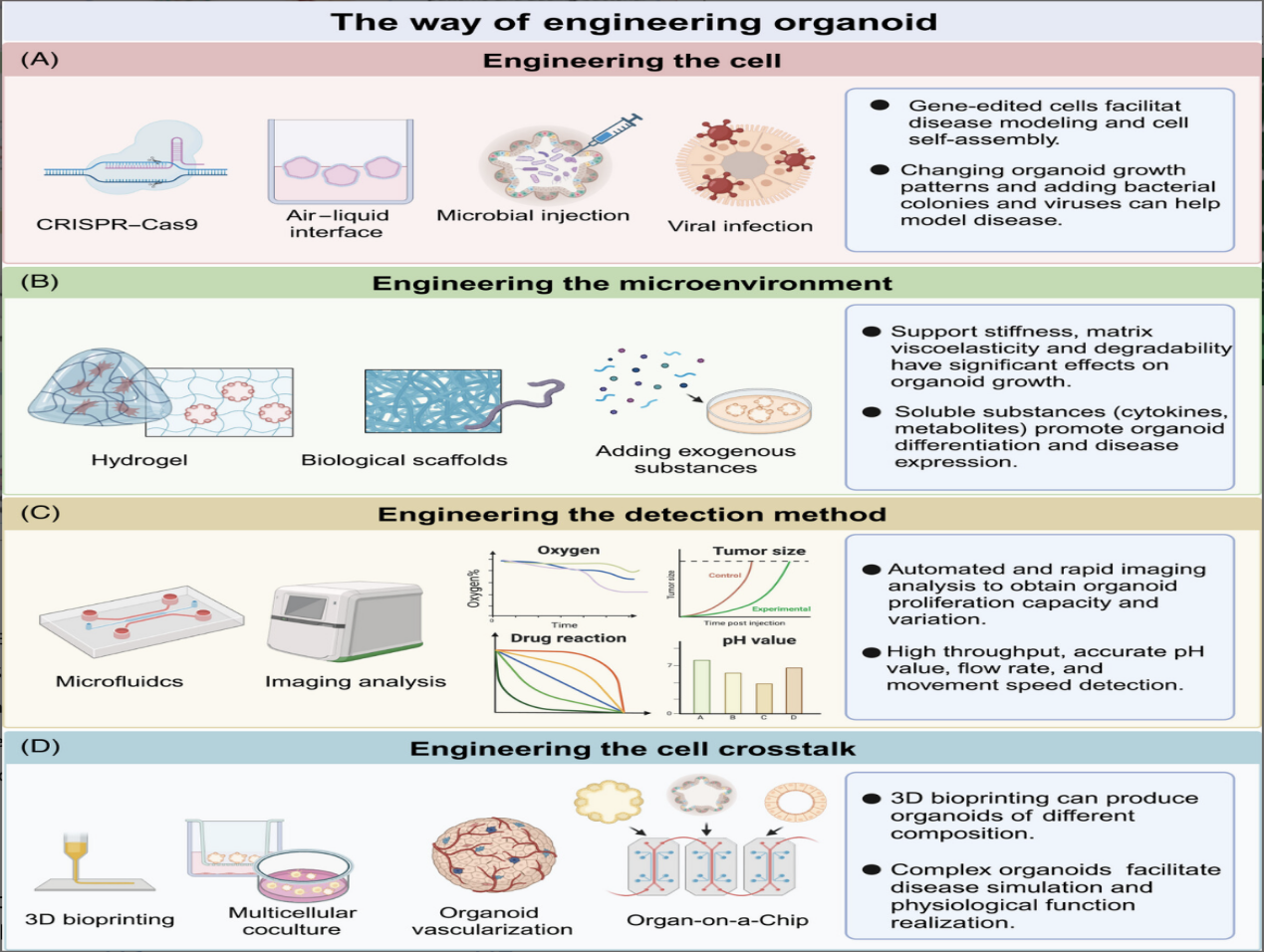
Applications of Organoids in Toxicity Prediction
1. Liver Toxicity
Drug-induced liver injury is a major cause of drug withdrawal. Liver organoids exhibit cytochrome P450 activity, bile canaliculi-like structures, metabolic zonation, and long-term functionality, enabling detection of idiosyncratic hepatotoxic responses not observed in animal systems (Karabicici et al., 2023).
2. Kidney Toxicity
Kidney organoids recapitulate nephron-like compartments including podocytes and proximal tubules, facilitating evaluation of filtration and tubular toxicity. These systems accurately reproduce injury patterns induced by nephrotoxic drugs such as cisplatin and aminoglycosides (Matsui & Shinozawa, 2021).
3. Cardiac Toxicity
Cardiac organoids simulate electrophysiological activity, contraction, mitochondrial function, and ion channel physiology. They identify arrhythmogenic compounds and cardiomyocyte stress mechanisms, particularly relevant for anti-cancer therapies (Matsui & Shinozawa, 2021).
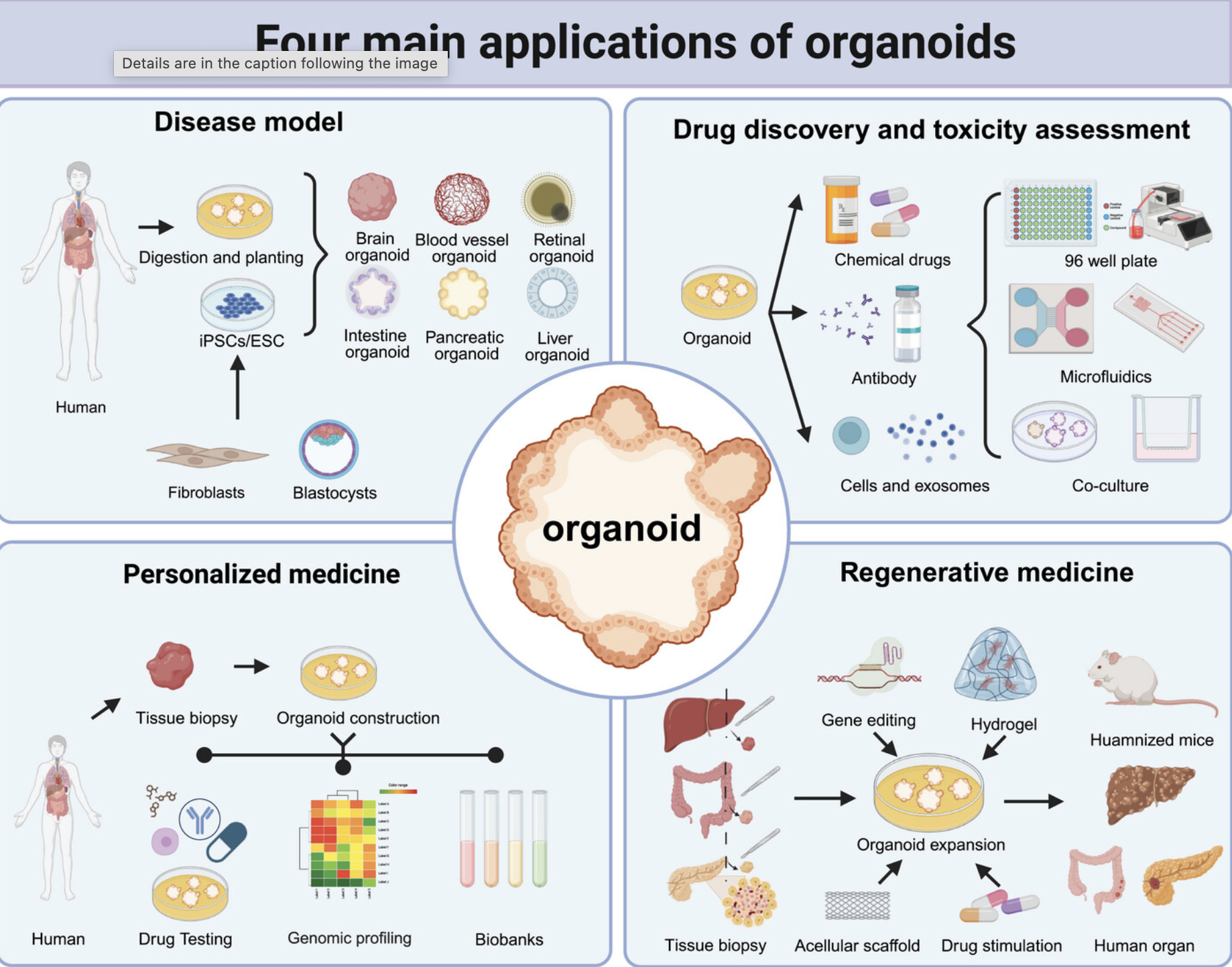
Advantages Over Traditional Models
Organoids offer several benefits over animal and 2D models:
Human-relevant metabolic and toxicological responses
Capability to model patient-specific and rare toxicities
Long-term culture for chronic toxicity assessment
Integration with organ-on-chip fluid dynamics for physiological exposure
AI-assisted high-throughput screening compatibility (Yao et al., 2024; Lee et al., 2025)
These features significantly enhance predictive power and reduce dependency on animals.
Challenges and Future Directions
Despite significant progress, organoids face challenges including limited vascular and immune integration, variability in culture protocols, incomplete maturation, and high production costs (Matsui & Shinozawa, 2021; Yao et al., 2024). Standardized quality control frameworks, scalable biomanufacturing systems, and regulatory validation guidelines are essential for full transition toward organoid-based safety testing. Future directions include vascularized and immune-competent organoids, automated bioreactor platforms, patient-derived organoid libraries, and AI-integrated toxicity profiling pipelines (Yao et al., 2024). Integration of multi-organoid systems is expected to enable whole-body pharmacokinetic modeling.
Conclusion
Organoids represent a transformative shift in drug toxicity assessment by providing human-relevant physiological responses, enabling the prediction of complex toxic events, and reducing reliance on animal testing. With ongoing advances in engineering, bioprocessing, and regulatory acceptance, organoids are poised to become a central component of next-generation preclinical evaluation pipelines.

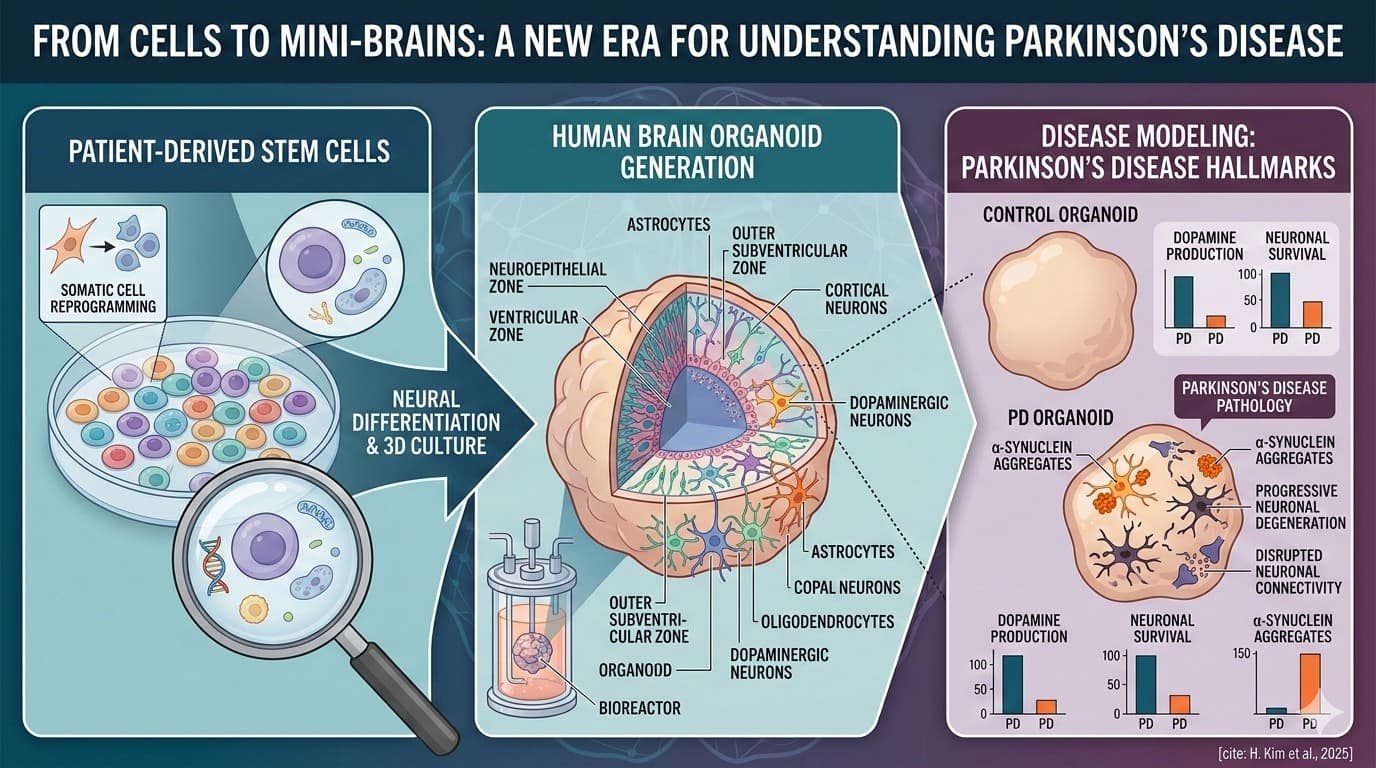


Discussion