Accuracy, noise, and information in cellular signalling
Cells constantly sense signals from their environment and convert them into internal actions through biochemical signalling pathways. Because only small numbers of molecules are involved and chemical reactions are inherently random, this process is naturally noisy, so even genetically identical cells can respond differently to the same stimulus. Despite this variability, many signalling systems achieve high precision and allow cells to reliably control gene expression, growth, and behaviour. Importantly, this precision is not free: improving signalling accuracy usually requires increased molecular activity, tighter regulation, and additional feedback mechanisms, all of which consume energy.
A signalling pathway can therefore be viewed as a communication system in which an external input, such as a hormone or ligand, is transformed into an internal output, such as gene expression or changes in cell shape, and its performance is better described by how reliably the response reflects the stimulus rather than by the average response alone. This reliability is commonly quantified using Fisher information, which measures how sensitively cellular responses change with small variations in the signal, and mutual information, which captures the overall statistical relationship between inputs and outputs across different signal levels. In essence, both measures express the same principle: signalling is more accurate when small changes in the input lead to clear and reproducible changes in the cellular response.
The yeast pheromone MAPK pathway as a quantitative model system
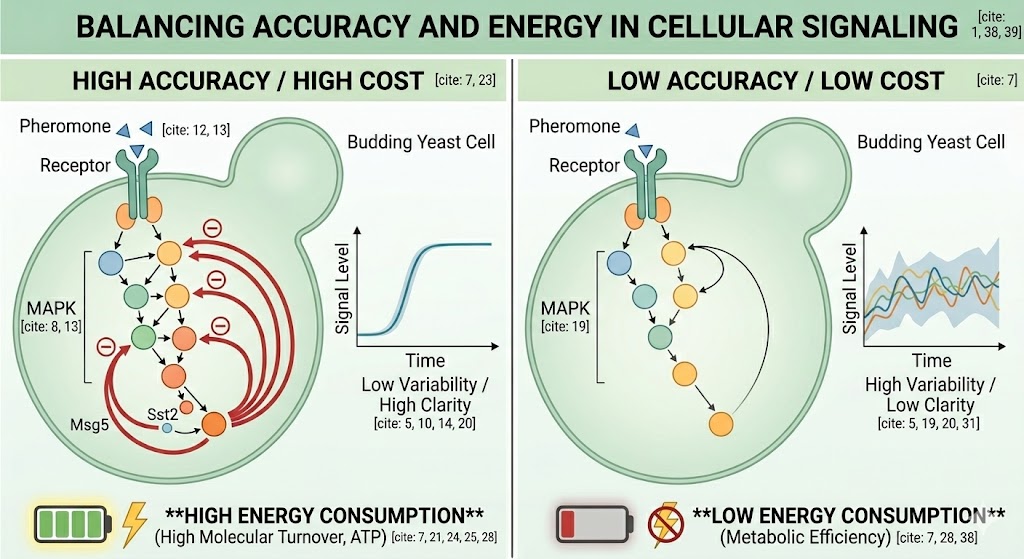
To investigate how real signalling networks balance accuracy and cost, a study (Anders, 2020) focuses on the pheromone response pathway of budding yeast, a well-established model for quantitative systems biology. In this pathway, extracellular mating pheromone activates a mitogen-activated protein kinase (MAPK) cascade that ultimately controls transcriptional programs and morphological changes associated with mating. Importantly, the pathway produces a highly coordinated and uniform morphological response, indicating that the underlying signal processing is both reliable and precise. At the same time, the network contains multiple regulatory interactions, including positive and negative feedback loops acting at different levels of the cascade. This combination of experimental accessibility, single-cell readouts, and rich regulatory architecture makes the yeast pheromone MAPK pathway an ideal system for analysing how network design shapes information transmission in living cells.
Negative feedback improves signalling accuracy.
In the yeast pheromone signalling pathway, two transcriptionally induced negative feedback regulators act at different levels: Sst2 functions upstream at the receptor–G protein step, while Msg5 acts downstream by dephosphorylating the MAPK Fus3. Each regulator controls distinct sources of variability within the signalling network. Experimental evidence shows that removing either feedback increases basal and low-stimulus activity and amplifies cell-to-cell variability. Overall, negative feedback suppresses background signalling and reduces molecular noise, thereby expanding the dynamic range and improving the accuracy of pheromone signal encoding.
Energetic constraints and fitness costs limit the optimisation of signalling accuracy.y
By combining mathematical modelling with targeted genetic experiments in the yeast pheromone pathway, Anders et al. (2020) showed that increasing the sensitivity of negative feedback—defined as how easily a feedback regulator is induced by pathway activity—can improve signalling accuracy, because earlier activation of the downstream regulator Msg5 reduces basal pathway activity and cell-to-cell variability, particularly at low stimulus levels, allowing cells to better discriminate small differences in pheromone concentration. However, this improvement is constrained by energetic costs, since feedback regulation requires continuous molecular turnover: upstream feedback via Sst2 consumes energy through repeated G-protein activation, while downstream feedback via Msg5 consumes energy through MAPK phosphorylation–dephosphorylation cycles.
Computational analysis further revealed that higher feedback sensitivity forces the pathway to operate at higher reaction fluxes to maintain the same output, thereby dissipating more energy, and that this cost strongly depends on feedback position, being modest for upstream feedback but much larger for downstream feedback. Crucially, the biological relevance of this energetic burden was demonstrated using a competition experiment that isolated the cost of MAPK phosphorylation cycles by comparing two inactive Fus3 variants, one capable of being phosphorylated and dephosphorylated and the other unable to enter this cycle; when grown together in the presence of pheromone, cells undergoing continuous enzymatic cycling showed a small but reproducible growth disadvantage, showing that although increasing feedback sensitivity enhances information transmission, the associated energy consumption imposes a measurable fitness cost and explains why signalling networks balance accuracy with metabolic efficiency rather than maximising information transmission.
New ways to understand how cells transmit information efficiently
Recent work by Tang and Hoffmann (2022) shows how modern data analysis and machine-learning tools can help scientists better measure how much information cells transmit through their signalling pathways, even when responses are noisy and change over time. Instead of looking only at simple input–output relationships, their review highlights that the dynamics of signalling, how signals evolve over time inside a cell, can also carry important information. This means that cells may use the shape and timing of their responses, not just their final level, to better distinguish between different stimuli, and that new computational methods can improve how precisely we study these processes.
Other recent theoretical studies (Saxena & Sharma, 2025) of the MAPK pathway further extend the work of Anders and colleagues by showing that the trade-off between signalling accuracy and energy use also depends on the basic physical properties of the reactions themselves. By analysing pathways with reversible reactions and fluctuating inputs, these studies show that both information transmission and energy consumption are strongly influenced by reaction thermodynamics, such as how much energy is lost during molecular cycles. Together, these results indicate that optimal cellular signalling is not only controlled by feedback strength and network design, but also by fundamental physical limits that link information, noise, and energy.
Conclusion
Overall, cellular signalling pathways are not optimized to maximize information, but to balance reliable signal transmission with the energetic and physical costs required to sustain it. Both feedback regulation and signal dynamics improve accuracy, yet fundamental thermodynamic and metabolic constraints ultimately limit how precisely cells can sense and respond to their environment.


Discussion