Understanding the human brain remains one of the greatest challenges in modern science. With billions of neurons forming intricate networks and constantly changing connections, the brain’s complexity makes it extremely difficult to study directly. For decades, neuroscientists have relied on animal models and two dimensional cell cultures to investigate how the brain develops and how neurological diseases emerge. While these approaches have provided valuable insights, they often fail to fully reproduce the unique biology of the human brain. In recent years, a new experimental system has begun to transform neuroscience research: human brain organoids.
What Are Brain Organoids?
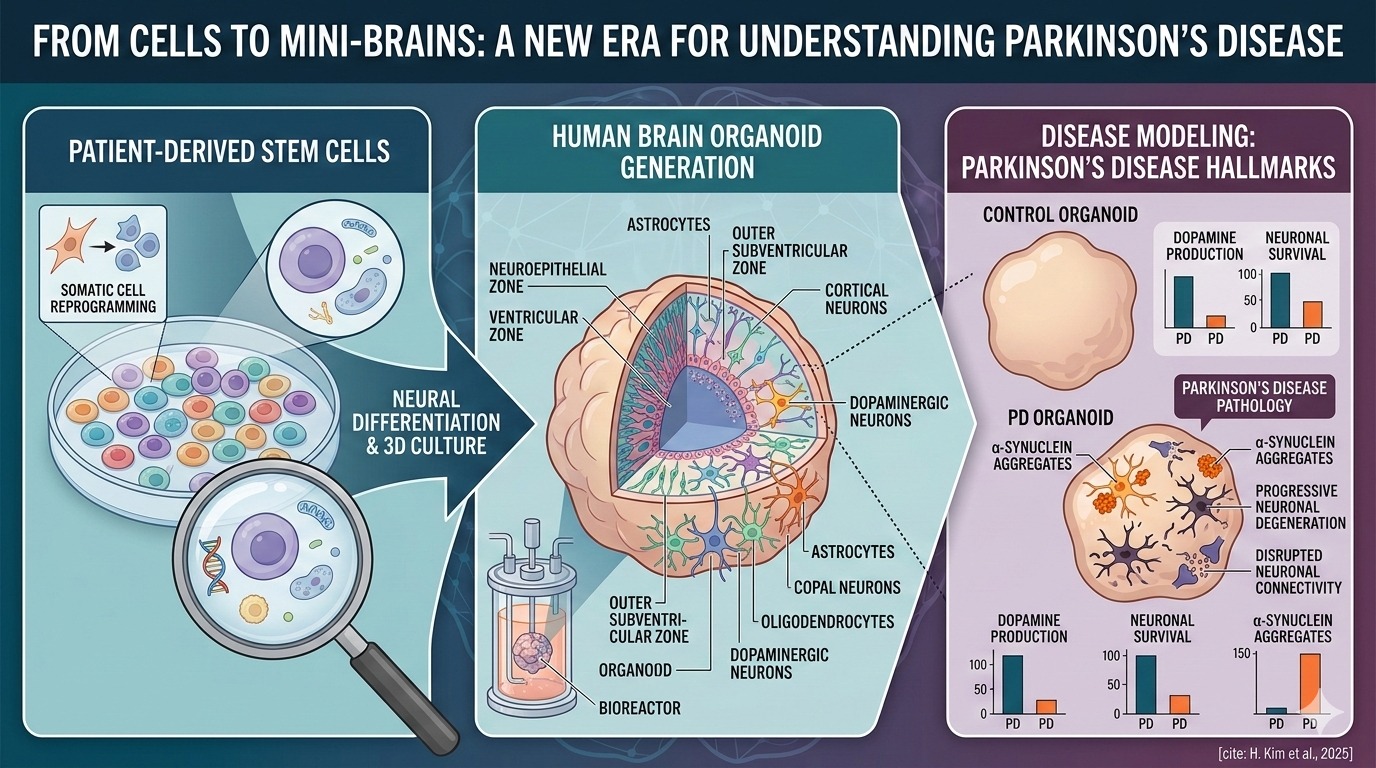
They are three-dimensional cellular structures grown in the laboratory from human pluripotent stem cells. Under controlled culture conditions, these cells can self-organize and differentiate into multiple neural cell types, including neurons and glial cells, forming tissue that resembles early stages of human brain development. Unlike traditional two-dimensional cell cultures, organoids develop spatial organization and cellular diversity that more closely reflect the architecture of the brain. Although they are far from complete or functional brains, these miniature models reproduce key biological processes, allowing researchers to study neural development, cellular interactions, and disease mechanisms in a human-specific context that was previously difficult to replicate experimentally.
Why Scientists Need Better Brain Models?
Developing effective treatments for neurological disorders depends heavily on the quality of the experimental models used to study them. Historically, much of our understanding of brain diseases has come from animal research, particularly studies in rodents. While these models have been invaluable for uncovering basic biological mechanisms, they often fail to capture the full complexity of human neurological conditions. Many molecular pathways, patterns of brain development, and disease progressions differ significantly between species. As a result, therapies that show promising results in animals frequently fail during clinical trials in humans.
Traditional cell cultures also present limitations, as growing neurons in flat, two-dimensional environments does not reflect the highly organized and interactive structure of living brain tissue. These challenges have created a significant gap between laboratory discoveries (H. Li et al., 2025). and successful medical treatments (Kim & Chang, 2023). To bridge this gap, researchers are increasingly turning to advanced in vitro systems capable of better representing the cellular diversity, organization, and dynamics of the human brain.
Modeling Brain Diseases with Organoids
One of the most powerful applications of brain organoids lies in their ability to reproduce key features of neurological diseases in a controlled laboratory environment. By generating organoids from patient-derived stem cells, researchers can recreate aspects of an individual’s genetic background and observe how disease-related processes unfold over time. This approach has opened new possibilities for studying disorders such as Alzheimer’s disease, Parkinson’s disease, autism spectrum disorders, and certain forms of epilepsy.
Within these miniature neural tissues, scientists (Kim & Chang, 2023) have been able to detect pathological hallmarks, including abnormal protein accumulation, disrupted neuronal connectivity, and progressive neuronal degeneration, that resemble those observed in patients. Because organoids develop in three dimensions and contain multiple interacting cell types, they allow researchers to examine how different cells contribute to disease progression. In addition, these systems provide a valuable platform for testing experimental drugs and identifying molecular pathways that may serve as targets for future therapeutic interventions.
Parkinson’s Disease
Recent research (H. Kim et al., 2025) has shown how organoid technology can help scientists study Parkinson’s disease, a neurological disorder in which specific brain cells produce dopamine gradually. In laboratory experiments, researchers created small three-dimensional brain-like structures that contain these dopamine-producing neurons. To do this, they converted ordinary human skin cells into neurons and allowed them to grow together in a 3D environment, forming miniature neural tissues that can be used to study how the disease develops. These structures reproduce several pathological features associated with Parkinson’s disease, including the accumulation of α-synuclein aggregates and progressive neuronal damage.
By incorporating supporting cells such as astrocytes into these systems, scientists can also examine how different cell populations influence neuronal survival or degeneration. Such models provide a valuable platform for exploring disease mechanisms and evaluating potential therapeutic strategies.
Current Limitations
Despite their remarkable potential, brain organoids still present several technical and biological limitations that researchers are actively working to overcome. One major challenge is the absence of vascular systems, which restricts nutrient and oxygen diffusion and can lead to cellular stress in deeper regions of the tissue.
In addition, organoids often display incomplete maturation, meaning that many cells resemble those found in early developmental stages rather than in fully developed adult brains. The variability between organoids generated in different laboratories also complicates reproducibility and standardization. Addressing these limitations will be essential to fully harness the potential of organoid-based models for studying complex neurological conditions.
Future Perspectives
As organoid technology continues to evolve, researchers are developing strategies to improve their biological realism and experimental reliability. Advances such as vascularized organoids, integration with gene-editing tools, and the incorporation of additional cell types, including immune and vascular cells, are expected to enhance their physiological relevance. These improvements could enable more accurate modeling of complex neurological disorders and accelerate the discovery of new therapeutic approaches.
Although brain organoids are still simplified representations of human neural tissue, they have already begun to transform neuroscience research. By bridging the gap between traditional laboratory models and human biology, organoids may play a crucial role in shaping the future of neurological disease research and treatment.


Discussion