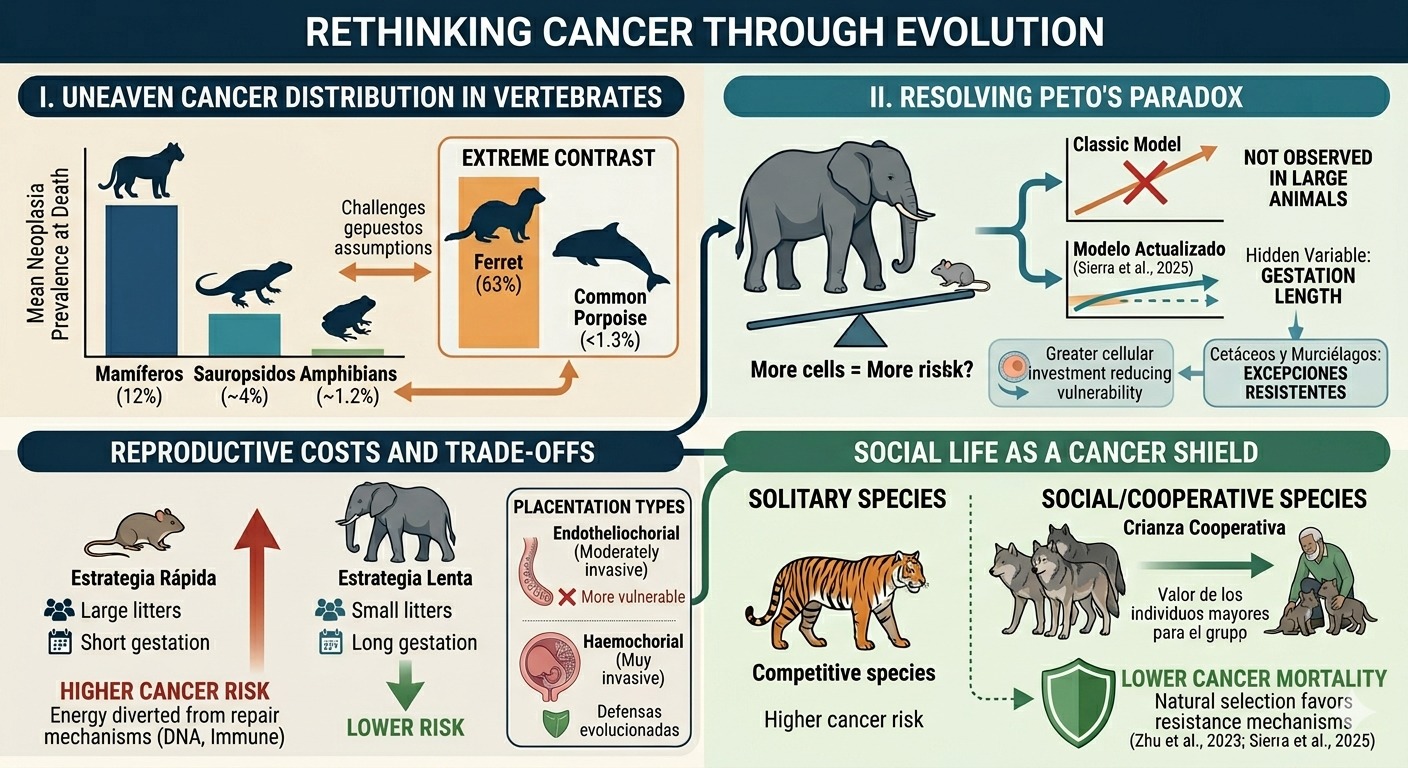
Why do some mammals develop cancer far more frequently than others? In ferrets, cancer prevalence exceeds 63%, while in the common porpoise it remains below 1.3%. This striking contrast challenges the assumption that cancer risk is primarily determined by genetics alone. Instead, emerging evidence suggests a more complex picture shaped by multiple biological and ecological factors, including body size, gestation length, reproductive strategies, and social behavior.
The field of comparative oncology is beginning to address these questions by systematically examining cancer prevalence across a wide range of species. As these patterns come into focus, they are offering new insights into the evolutionary dynamics of cancer and prompting a reassessment of how susceptibility to disease is distributed across the animal kingdom.
Cancer is everywhere in the animal kingdom, but not equally
Cancer is a widespread biological phenomenon, not limited to humans, but its distribution across the animal kingdom is far from uniform. An analysis (Compton et al., 2024) of 16,049 necropsy records spanning 292 vertebrate species, including mammals, birds, reptiles, and amphibians, revealed that all major taxonomic groups experience cancer. However, prevalence differs markedly: mammals exhibit the highest median rate of neoplasia at death (12%), followed by sauropsids (~4%) and amphibians (~1.2%). Notably, ferrets (Mustela putorius) show exceptionally high rates, with 63% affected, often by lymphoma, whereas species such as the Rodrigues fruit bat remain below 1.6%, underscoring cancer’s uneven burden across life.
Size, lifespan, and the paradox that launched a field
At first glance, cancer risk should scale with size and longevity: more cells dividing over longer lifespans ought to increase the probability of malignant transformation. This expectation, known as Peto’s paradox, is contradicted by observations (Compton et al., 2024) in large animals such as elephants, which do not exhibit exceptionally high cancer rates. For decades, comparative studies failed to detect a consistent relationship between body mass and cancer prevalence across species.
Recent large-scale analyses (Sierra et al., 2025) help resolve this paradox. When gestation length is taken into account, a modest positive association between body size and cancer emerges. Gestation appears to act as a hidden variable: species with longer developmental periods may invest more in cellular maintenance, reducing vulnerability. Notably, cetaceans and bats remain outliers, combining large size or long lifespans with enhanced cancer resistance.
Reproduction and cancer, a costly trade-off
Reproduction is one of the most energy demanding processes in biology, and its costs may extend beyond immediate survival to influence cancer risk. Comparative studies (Dujon et al., 2023) show that species producing larger litters tend to exhibit higher cancer prevalence and mortality. This pattern reflects fundamental life-history trade-offs: energy allocated to reproduction may reduce investment in DNA repair, immune surveillance, and other protective mechanisms against malignant transformation.
Gestation length further reinforces this relationship. Species with shorter gestation periods, typically associated with fast reproductive strategies, display higher cancer mortality, whereas longer gestation correlates with reduced risk. Interestingly, placentation type adds nuance to this picture. Species with highly invasive haemochorial placentation do not show the highest cancer mortality, likely due to evolved defenses. Instead, those with moderately invasive endotheliochorial placentation appear most vulnerable, suggesting incomplete evolutionary compensation against cancer risk.
Social Life as a Cancer Shield
Behavior, not just biology, plays a critical role in shaping cancer risk. Species that live in groups, reproduce cooperatively, and exhibit longer gestation periods tend to show lower cancer mortality than solitary species, even after controlling for diet. Social carnivores such as wolves and lions, for example, do not display the elevated cancer rates observed in solitary counterparts.
This pattern reflects an evolutionary logic: in cooperative systems, older individuals contribute to offspring survival, increasing their value to the group. As a result (Zhu et al., 2023 & Sierra et al., 2025), natural selection may favor mechanisms that reduce cancer risk in these individuals. In contrast, in more competitive species, the loss of older individuals may not be detrimental, and can even enhance population dynamics, allowing cancer susceptibility to persist.
So What?
What do these cross-species patterns mean for us? First, they point to natural strategies of cancer prevention. Traits such as long gestation, slow life histories, and cooperative social systems appear linked to enhanced cancer resistance. Uncovering the molecular basis of these traits, ranging from expanded tumor suppressor gene families to improved DNA repair and immune regulation, could inform new approaches in human prevention and therapy.
Second, certain species may offer more realistic models of cancer. Animals like opossums and hedgehogs develop spontaneous tumors that resemble human disease more closely than engineered laboratory models.
Finally, these findings reshape how we think about cancer itself. Rather than a simple consequence of chance or cellular failure, cancer emerges as a condition influenced by evolutionary pressures. Across species, its risk reflects how organisms grow, reproduce, and interact, suggesting that evolution has not only constrained cancer, but in some contexts, tolerated it.



Discussion