Introduction
Imagine a world where tiny scaffolds quietly support every building, keep doors in the right place, and prevent chaos from spreading. In our bodies, that silent scaffolding exists too, and it’s called the basement membrane. This ultra-thin layer sits beneath epithelial cells only on the basal side, the cells that line organs, skin, and blood vessels, providing both structure and protection. Though often overlooked, the basement membrane is indispensable: they act as a selective barrier. Damage or malfunction in this tiny layer can trigger diseases ranging from kidney failure to muscular dystrophy, cancer metastasis, and neurodegenerative disorders like Alzheimer’s disease. Basement membrane orchestrates complex interactions that keep tissues healthy. Scientists are only beginning to uncover the secrets of the basement membrane. What we’ve learned so far reveals a story of tiny structures with colossal influence — a perfect example of how small things can make a huge difference in life and health.
The Basement Membrane: A Living Molecular Mesh
If cells were cities, the basement membrane would be their pavement, plumbing, zoning laws, and Wi‑Fi—all rolled into one impossibly thin layer. Long thought to be little more than biological wallpaper, the basement membrane is now understood as a dynamic, information‑rich structure whose composition is as carefully tuned as a symphony orchestra.
At its core, every basement membrane is built from a surprisingly small set of molecular ingredients, but their organization creates astonishing diversity. The four principal structural components are laminins, type IV collagen, nidogens, and heparan sulfate proteoglycans, particularly perlecan and agrin. These molecules are secreted as soluble building blocks that self‑assemble outside the cell into a resilient, nanoscale sheet.
Laminins act as the master architects of this structure. These large, multi‑armed proteins attach directly to cell‑surface receptors and initiate basement‑membrane formation. Without laminins, other components fail to assemble properly—a fact underscored by genetic studies showing that loss of laminins causes catastrophic failure of early embryonic development.
Once the laminin framework is established, type IV collagen provides strength. Unlike the rigid, rope‑like collagens found in tendons or bone, collagen IV forms an interwoven molecular network that is both tough and flexible. This architecture allows basement membranes to withstand mechanical stress while remaining permeable — essential for tissues such as kidney filters, blood vessels, and muscle.
Nidogens function as molecular connectors, binding both laminin and collagen IV, effectively stitching their networks together. While not always essential for initial assembly, nidogens lend stability and resilience to the mature basement membrane.
The final key components, perlecan and agrin, transform the basement membrane from a structural scaffold into a biochemical command center. These large proteoglycans are decorated with long sugar chains that bind growth factors, cytokines, and morphogens. In doing so, the basement membrane becomes a reservoir and regulator of cellular signals, controlling when and where cells divide, migrate, or specialize.
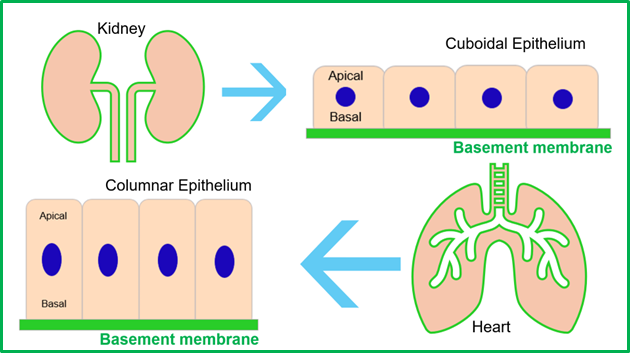
When the Shield Breaks: Diseases Linked to Basement Membranes
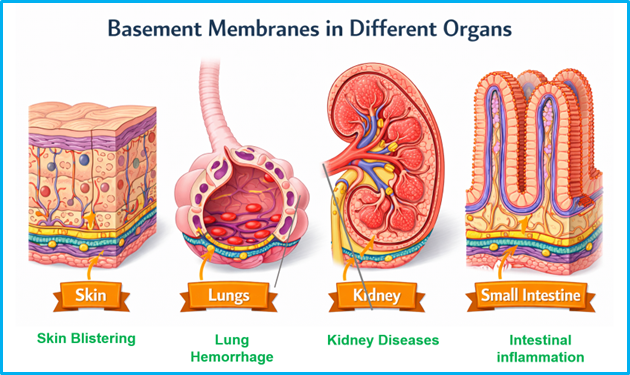
Basement membranes act as protective shields for organs, and when they fail, the consequences can be severe. Consider the kidney, where the basement membrane forms the glomerular filter. When collagen IV is mutated, this filter becomes leaky, leading to chronic kidney diseases like Alport syndrome. Patients can suffer proteinuria, hematuria, and progressive kidney failure. In the skin, basement membrane disruption results in blistering diseases, where minor friction can tear tissue layers apart. In the lungs, defective basement membrane can cause hemorrhage, while in the gut, basement membrane damage may contribute to inflammatory conditions. Even the eyes rely on an intact basement membrane for lens and retina stability, so vision problems can emerge when the basement membrane is compromised.
The underlying principle is simple: the basement membrane is a selective barrier and organizer. When its structure is altered, cells can lose their orientation, signaling goes awry, and tissues weaken. Cardiovascular diseases are linked to basement membrane abnormalities in blood vessels, while neurodegenerative disorders like Alzheimer’s show thickened or dysregulated basement membranes around neurons. These changes contribute to impaired nutrient flow, waste clearance, and inflammatory responses in the brain.
What’s striking is how often basement membrane -related dysfunction remains invisible until a disease manifests. Unlike a broken bone, you can’t see basement membrane damage, but its ripple effects affect multiple organs. Think of it as a dam: even a tiny crack can flood entire towns downstream. By studying basement membrane defects, scientists are uncovering mechanisms behind a broad spectrum of conditions from congenital syndromes to age-related organ degeneration, revealing that this tiny layer is vital for maintaining life’s delicate balance.
Mutations That Matter: The Genetic Side of Basement Membranes
The genetic story of basement membranes shows that small changes in DNA can have massive consequences. Each basement membrane protein is encoded by specific genes, and mutations can dramatically alter tissue function. For example, collagen IV mutations often compromise kidney filtration, causing Alport syndrome. Patients experience progressive kidney failure, hearing loss, and sometimes ocular problems. Laminin mutations affect muscle integrity, leading to muscular dystrophy. Without laminin, muscle fibers cannot properly attach to their basement membrane, making them prone to damage and degeneration.
Mutations in Agrin, a basement membrane component at the neuromuscular junction, can cause congenital myasthenic syndromes, where patients struggle with muscle weakness and fatigue because nerve signals fail to effectively stimulate muscles. Similarly, defects in Perlecan disrupt skeletal development and neuromuscular function, emphasizing the critical role of the basement membrane in coordinating tissue architecture.
These genetic insights highlight a broader principle: the basement membrane is not just structural scaffolding, but a molecular linchpin connecting genes, proteins, and cellular behavior. A single mutation can propagate through organ systems, affecting skin, kidneys, muscles, and nerves simultaneously. Scientists are beginning to map these cascading effects, revealing complex networks of interaction where basement membrane proteins act as both building blocks and signaling hubs. It’s a vivid reminder that in biology, even the tiniest molecular misstep can trigger dramatic consequences across the body.
Basement Membranes and Cancer: When the Scaffold Turns Rogue
In cancer, basement membranes are no longer just silent scaffolds — they can become unwitting accomplices in disease progression. Normally, the basement membrane serves as a barrier, keeping epithelial cells in place and preventing them from invading neighboring tissues. But cancer cells are crafty. Primary tumor cells often alter the composition of the basement membrane, secreting proteins like laminin and modifying others to weaken the structural barrier. This remodeling creates “escape routes” for tumor cells, enabling them to invade nearby tissues and eventually travel to distant organs, a process known as metastasis.
In highly metastatic cancers such as breast, prostate, and colon tumors, tumor cells not only degrade the normal basement membrane but also actively secrete its components to engineer a microenvironment that supports their migration. Laminin overexpression, in particular, is closely linked with tumor aggressiveness. The same proteins that normally maintain tissue organization now assist cancer cells in spreading — a microscopic betrayal that highlights the dual nature of basement membranes.
Interestingly, the composition and structure of the basement membrane are not uniform; different organs and tissues show unique basement membrane arrangements, and cancer cells exploit these differences to optimize invasion. For instance, in metastatic breast cancer, laminin-rich regions form tracks along which tumor cells migrate more efficiently. Research has shown that basement membrane remodeling can also influence immune cell behavior, either masking tumor cells from detection or creating inflammatory signals that paradoxically support tumor growth.
Understanding the basement membrane's role in cancer has revolutionized the way scientists think about tumor progression. By targeting basement membrane remodeling, laminin overexpression, or basement membrane protein signaling, researchers hope to develop therapies that block metastasis at the very molecular level. This tiny layer, invisible without a microscope, proves that even the smallest structures can dictate the fate of life and death in our bodies — transforming a silent supporter into a major player in one of humanity’s deadliest diseases.
Aging, Open Questions, and the Ever-Changing Basement Membrane
Basement membranes are dynamic structures that change over time, and their age-related remodeling has profound consequences. As we grow older, basement membrane layers often thicken and stiffen, which can compromise tissue flexibility and cellular communication. In the kidney, this thickening reduces filtration efficiency, contributing to chronic kidney disease. In the brain, basement membrane alterations are observed in Alzheimer’s disease, where excessive deposition around blood vessels may impair waste clearance and nutrient delivery, exacerbating neuronal damage. Similarly, in multiple sclerosis, inflammatory processes trigger excessive basement membrane deposition, which can hinder nerve repair and amplify disease progression. Even in diabetes, basement membrane thickening in blood vessels contributes to organ damage, highlighting the widespread impact of age-related basement membrane remodeling.
Beyond structural changes, the basement membrane composition can shift with age. Certain proteins, like laminin and collagen IV, may be overproduced or underproduced, altering cell signaling and tissue homeostasis. These changes can make tissues more prone to injury, inflammation, and fibrosis. Scientists are also exploring how mechanical stress interacts with basement membrane remodeling, affecting organ resilience over decades.
Despite decades of research, many questions remain. How do cells sense subtle basement membrane changes and adapt? Can we manipulate basement membrane composition to prevent age-related diseases? Why do some tissues maintain functional basement membrane while others deteriorate rapidly? These mysteries make the basement membrane a frontier of modern biology.
For a structure only a few hundred nanometers thick, the basement membrane wields enormous influence over health, disease, and aging. Its dynamic nature, coupled with its vulnerability to mutation and stress, reveals a tiny layer with a massive role in our bodies’ resilience and longevity. Studying these changes not only illuminates fundamental biology but also opens doors to potential therapies targeting age-associated diseases and tissue degeneration.
Conclusion
Basement membranes are microscopic heroes of the body. These ultra-thin extracellular matrices provide structural support, guide cellular behavior, and act as essential barriers that protect organs from chaos. Damage, mutation, or remodeling of basement membrane proteins can trigger diseases ranging from kidney failure and muscular dystrophy to cancer and Alzheimer’s disease. Yet even as researchers uncover the secrets of these tiny layers, much remains to be discovered about their complex roles in development, aging, and disease. What is clear is that the basement membrane, though invisible to the naked eye, is indispensable — a tiny layer carrying a huge job. Recognizing its power reminds us that in biology, as in life, the smallest players often make the biggest difference.



Discussion