Introduction
Cells are like bustling cities, where tiny delivery trucks—called vesicles—transport essential cargo to keep everything running smoothly. The SNAP29 gene acts as the city’s famous dockmaster, ensuring every package reaches its proper destination. When the SNAP29 gene mutates, this system fails, and the rare genetic curse CEDNIK syndrome emerges. It attacks body and sense alike – shrinks the brain, steals eyesight and hearing, lashes the body with seizure and scaly skin. Life under CEDNIK syndrome is brief, fragile, and a daily struggle against a merciless genetic storm. This article explores how the SNAP29 gene controls trafficking inside of the cell, what happens when it malfunctions, and why understanding it matters for patients and science alike.
The City Inside Our Cells: never sleeps
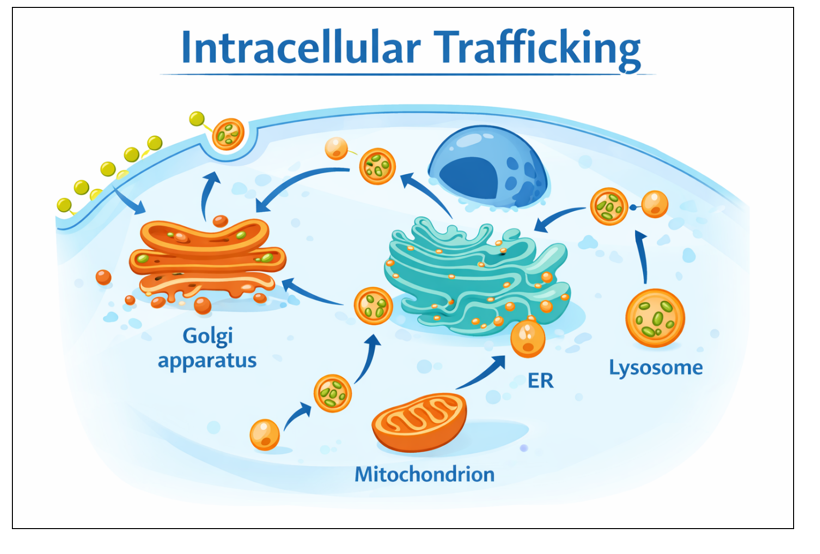
A cell is a microscopic city you cannot see, yet more organized than any other metropolitan city on earth. Inside every living cell is a bustling, never-sleeping city, alive with streets, highways, and delivery trucks called vesicles that carry the cargo of life -proteins, lipids, and enzymes – throughout the cell. These vesicles ensure that every part of the cell receives what it needs to function properly.
SNAP29: The Famous Dockmaster
Imagine a city where trucks never reach the right warehouses. The SNAP29 gene, located on chromosome 22, serves as the non-negotiable dockmaster, helping vesicles fuse with the Golgi, endosomes, and lysosomes so that cargo is offloaded correctly.[1] Without SNAP29, deliveries stall, and the city falls into chaos.
Critical Deliveries in the Skin
In the skin, special vesicles called lamellar granules carry lipids and enzymes to the outer layer, building a strong, protective barrier.[2] When the SNAP29 gene is mutated, these granules stall, leaving the skin scaly and thick.[3]
Critical Deliveries in the Brain and Nerves
Even beyond the skin, vesicles transport molecules critical for other tissues. SNAP29 ensures that nerve cells receive proteins needed to grow, laying the foundation for proper brain development and nerve communication.[1] When these deliveries fail, the consequences for the body can be profound, setting the stage for the symptoms seen in CEDNIK syndrome.[2]
When the Cellular City Falls into Chaos: CEDNIK Syndrome
When the SNAP29 gene is mutated, vesicles cannot fuse correctly, and essential cargo never reaches its destination. In nerve cells, these delivery trucks stall, leaving them hungry, so brain cells can’t grow or talk to each other properly. This leads to abnormal brain development, developmental delays, seizures, and nerve damage.
Communication Problems in the brain
Failed vesicle fusion also disrupts neurotransmitter release, preventing nerve cells from signaling effectively. The brain’s network becomes less efficient, and neurological complications accumulate.
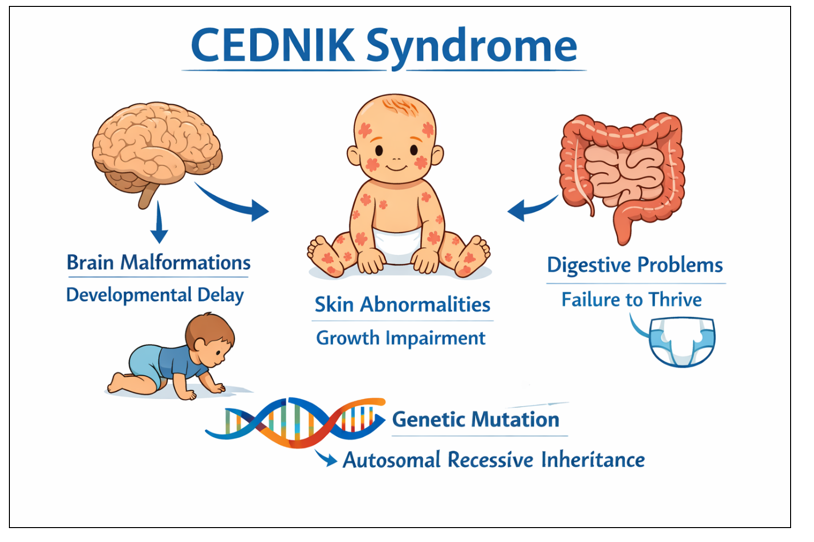
Symptoms of CEDNIK Syndrome
Patients with CEDNIK syndrome may experience:
Abnormal brain development
Developmental delays
Neuropathy and seizures
Scaly skin and thickened palms/soles
Impaired vision and hearing[1]
CEDNIK syndrome is an autosomal recessive disorder, meaning a child must inherit two copies of the mutated SNAP29 gene, one from each parent, to be affected. Diagnosis is usually suspected based on skin and neurological symptoms and confirmed through genetic testing or whole exome sequencing.[2]
Currently, there is no cure, and treatment is supportive, focusing on managing symptoms. Prognosis is generally poor due to the disorder’s rarity. Life expectancy varies: some patients pass away in early childhood from neurological complications or infection, while others survive into their late teens or early twenties.[3]
Practical Implications
Understanding how the SNAP29 gene keeps the cell’s delivery system running could help scientists design targeted therapies that restore health in people with CEDNIK syndrome.
Conclusion
CEDNIK syndrome shows how fragile our cellular cities can be: a single faulty dockmaster, the SNAP29 gene, can bring vital deliveries to a halt. When cargos fail to reach their targets, the effects ripple across skin, nerves, and brain, causing severe symptoms and developmental delays. Studying the SNAP29 gene and its role in trafficking is more than basic science—it reveals how microscopic disruptions can shape entire lives. By understanding these cellular traffic jams, researchers hope to develop future treatments, giving hope to patients with rare genetic disorders like CEDNIK syndrome.



Discussion