The Great Biological Wall
For decades, treating brain diseases has been like trying to deliver a vital letter to a fortress that refuses to open its gates. The blood–brain barrier (BBB) stands as one of the most sophisticated protective systems in human biology, sealing the central nervous system behind a tightly regulated cellular wall formed by endothelial cells, reinforced by astrocytes and pericytes, and guarded by powerful molecular “bouncers” such as efflux transporters.
This architecture is essential for shielding neurons from toxins, pathogens, and fluctuations in the bloodstream, but it also creates a devastating bottleneck for modern medicine. More than 98% of small-molecule drugs and nearly all large biological therapeutics fail to cross the BBB in meaningful amounts, leaving many promising treatments for neurodegenerative disorders, brain tumors, and inflammatory diseases stranded in the circulation. As highlighted by recent work (Li et al., 2024) on exosomes in central nervous system disorders, the problem is not simply physical obstruction: it is an active, dynamic barrier that senses, filters, and expels foreign compounds with remarkable efficiency, particularly under pathological conditions where inflammation and vascular dysfunction further distort drug delivery.
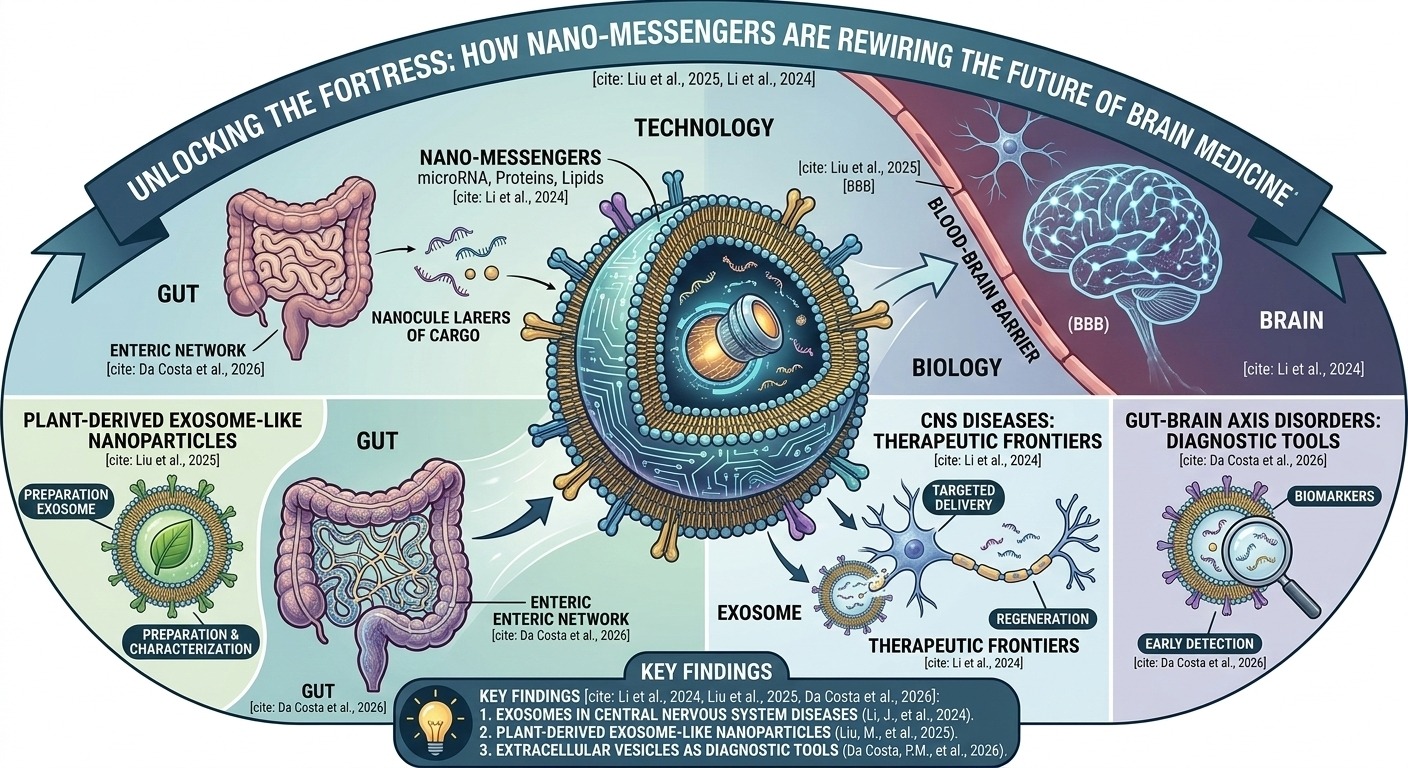
Yet within this heavily guarded environment, the brain already relies on its own nanoscale communication network to exchange information between neurons, glia, and distant cells. These natural nano-messengers, exosomes, circulate across biological boundaries that most synthetic carriers cannot penetrate, suggesting a radical shift in strategy: instead of forcing drugs through the gates, what if we could disguise our therapeutic messages as part of the brain’s own internal mail system, and let biology itself show us the way in?
The nano-messengers
Exosomes are tiny, bubble-like particles released by our own cells that work as a highly efficient biological delivery system. Thousands of times smaller than a human cell, exosomes are surrounded by a protective lipid membrane and carry carefully selected molecular “packages”, including proteins, fats, and different types of RNA, that allow one cell to influence the behavior of another.
For many years, scientists believed these particles were nothing more than cellular waste. Today, we know that exosomes are deliberately produced through an organized internal pathway. Inside the cell, small membrane compartments form and mature into structures known as multivesicular bodies, which act like sorting hubs. From there, selected molecules are packed into tiny internal vesicles and released outside the cell as exosomes.
What makes exosomes especially powerful is that their cargo is not random. Each cell loads information that reflects its current state, whether it is stressed, inflamed, healthy, or diseased. When another cell takes up these vesicles, the messages they carry can change gene activity, alter signaling pathways,s and even reshape how that cell responds to its environment.
In the brain, where direct cell-to-cell communication is tightly regulated, exosomes have emerged as a natural long-distance messaging system between neurons, glial cells, l,s and the surrounding vascular network. This biological “courier service” is now attracting intense interest in medicine because it offers a ready-made, highly compatible way to transport complex instructions through the body, including potentially therapeutic ones.
From Plant Exosomes to Liquid Biopsies of the Brain
One of the most intriguing discoveries in recent years is that not all useful exosomes have to come from human cells. Research on plant-derived exosome-like nanoparticles (PDELNs) (Liu et al., 2025) shows that tiny vesicles naturally released by edible plants can survive digestion, remain biologically stable, and interact with human cells. Even more surprisingly, these plant nanoparticles display very low immunogenicity and can be engineered to carry therapeutic molecules. This opens an entirely new and sustainable source of biological carriers, raising the possibility that future brain therapies could be delivered using biocompatible messengers that originate outside the human body yet are readily accepted by it.
At the same time, exosomes are emerging as powerful cross-organ messengers, particularly along the gut–brain axis. Recent studies (Da Costa et al., 2026) show that extracellular vesicles released by intestinal and immune cells can enter the bloodstream and reflect molecular changes associated with neurological and psychiatric conditions. Because these vesicles carry proteins and RNA signatures associated with brain-related dysfunction, a simple blood sample can act as a liquid biopsy, revealing aspects of brain health without directly accessing the brain. In this sense, exosomes are not only delivery vehicles but also biological reporters—linking the digestive system and the brain through a shared nanoscale communication network that may transform how mental and neurological disorders are detected and monitored.
Challenges and Hope
Despite their enormous promise, exosomes are not yet ready for routine clinical use. One of the main technical obstacles is isolating exosomes with high purity and consistency, which remains difficult, and each batch can contain vesicles of diverse sizes and molecular cargo. This biological heterogeneity makes it harder to standardize treatments and predict exactly how each preparation will behave within the human brain.
Yet, progress exists. New and more refined sorting strategies, such as improved ultracentrifugation protocols and emerging microfluidic platforms, are helping researchers separate, characterize, and engineer exosomes with far greater control. As these technologies mature, the vision that once seemed unrealistic, a non-invasive, targeted,d and biologically compatible therapy for diseases of the central nervous system, is steadily moving from an experimental concept toward a realistic clinical future.


Discussion