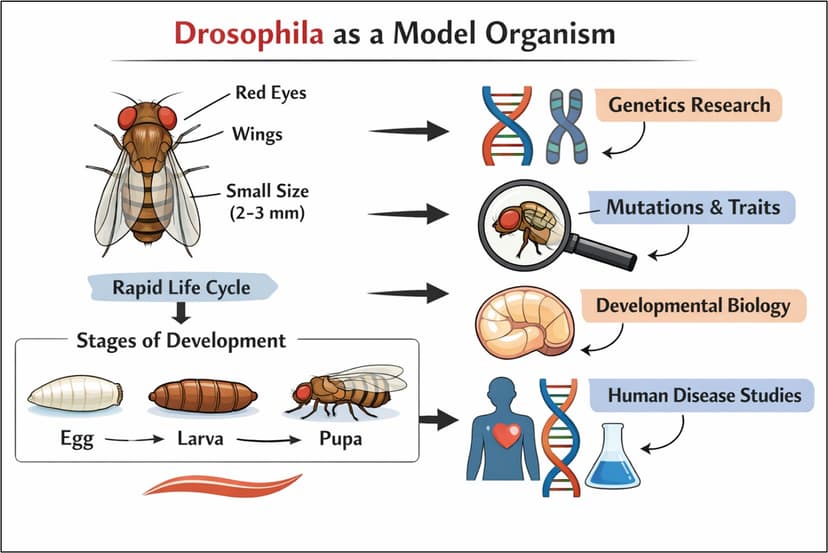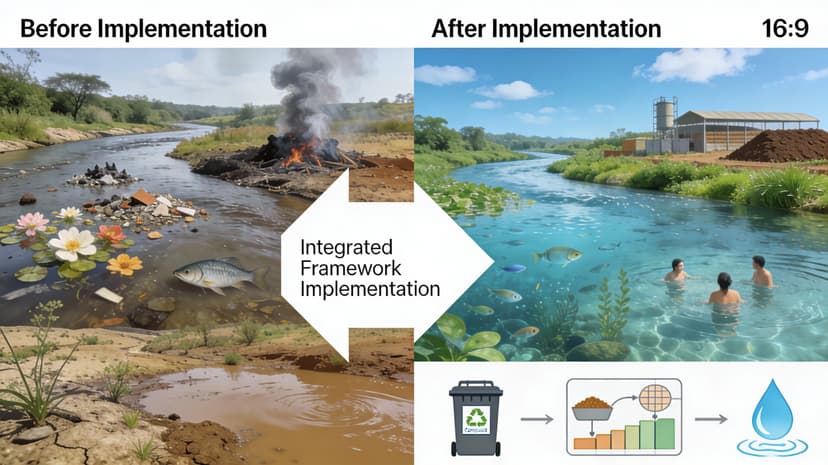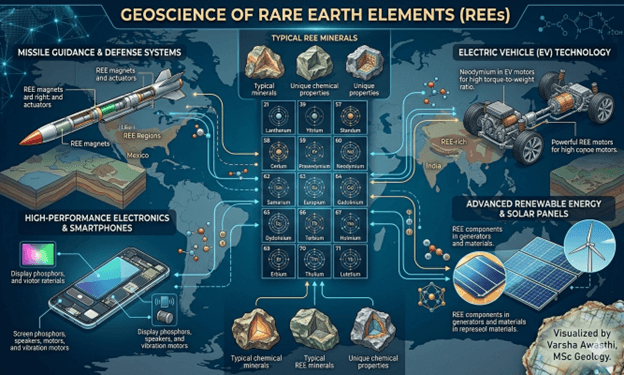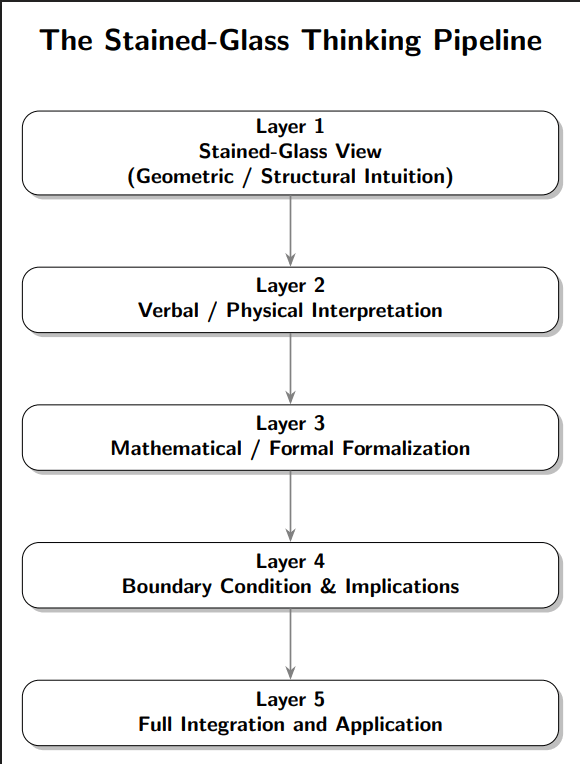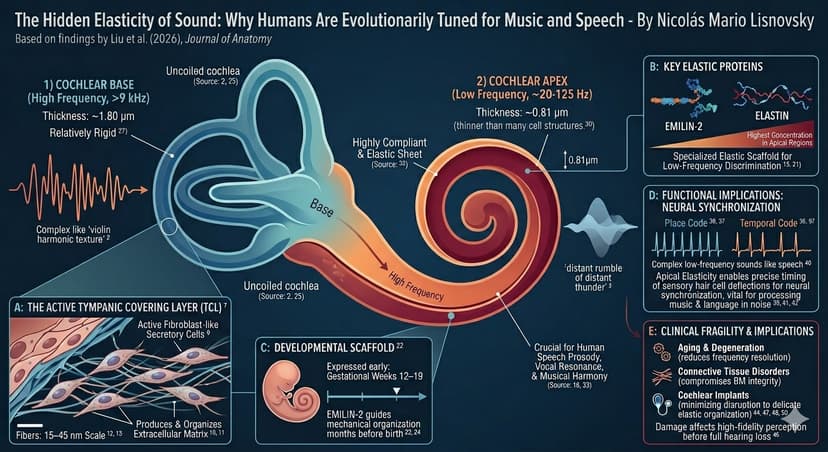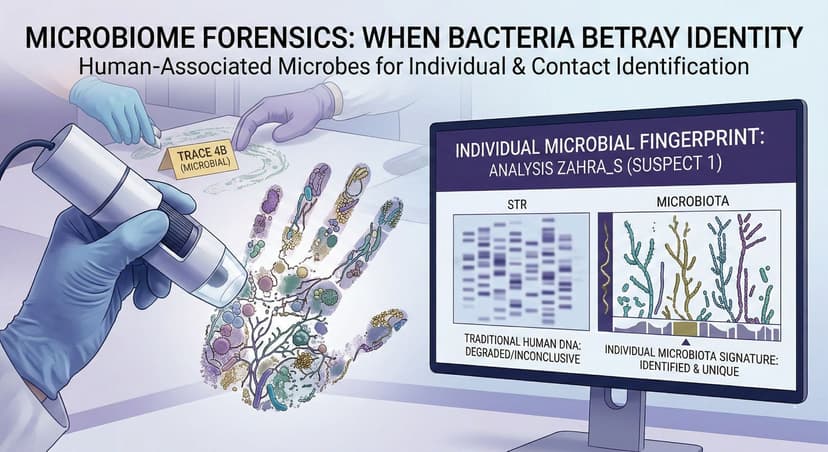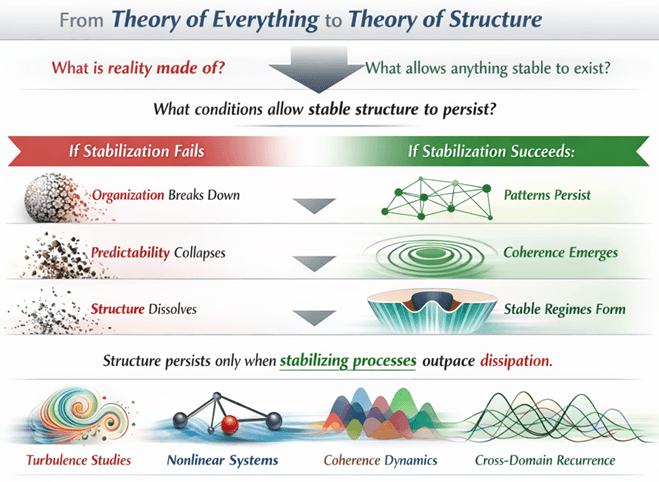
Why We Need Verification and Validation of Medical Devices
Title 21 Part 820 of the Code Of Federal Regulations (CFR) details the requirements for the design and production of medical devices. These strict, comprehensive requirements are in place for good reason, we want these devices to work.